Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4A: Endoscopy / Esophagus
61 - Impact of GLP-1RA Therapy on Esophageal Motility Patterns
Wednesday, October 29, 2025
9:30 AM - 9:40 AM PDT
Location: North Ballroom 120D

Annie L. Wang, BS, BA (she/her/hers)
Duke University School of Medicine
Durham, NC
Presenting Author(s)
Award: ACG Medical Student Outstanding Research Award
Annie L. Wang, BS, BA1, Laura Bach, DO2, David A. Leiman, MD, MSHP3
1Duke University School of Medicine, Durham, NC; 2Duke University, Durham, NC; 3Duke Clinical Research Institute, Durham, NC
Introduction: Glucagon-like peptide-1 receptor agonist (GLP-1RA) use is associated with delayed gastric emptying. It is unknown, however, whether GLP-1RAs affect esophageal motility. We aimed to assess the relationship between GLP-1RA use and esophageal function test results.
Methods: Patients with type 2 diabetes undergoing high-resolution manometry (HRM) between December 2022 and October 2024 were retrospectively evaluated, comparing cases with controls (on versus off GLP-1RA). HRM diagnoses per Chicago Classification 4.0, reflux testing results, and patient-reported symptom scores from validated questionnaires including the Brief Esophageal Disease Questionnaire, Eckardt, and Reflux Disease Questionnaire were collected. Patients with prior foregut surgery and known conditions associated with dysmotility (e.g., scleroderma) were excluded. Comparisons were performed using Chi-square and Mann-Whitney U tests for categorical and continuous measures, respectively.
Results: There were 132 patients (n=42 cases) assessed and most (n=29, 69.0%) cases were female. Compared to controls, patients receiving GLP-1RAs were younger (median 62.5 vs 67.5 years, p=0.04) and had higher BMI (median 33.3 vs 30.7 kg/m2, p=0.01), but there were no differences in type 2 diabetes control or use of acid suppression medications between groups. Semaglutide was the most commonly used GLP-1RA (n=24, 57.1%) among cases, and there was no association (p=0.51) between GLP-1RA type or dose and esophageal motility diagnoses. Most GLP-1RA users (n=27, 64.3%) had a normal HRM, and there was no significant difference (p=0.071) in overall HRM diagnoses between cases and controls. When restricting analyses to patients who underwent HRM without concomitant pH testing, there remained no significant difference in HRM diagnoses (p=0.15). Among cases and controls undergoing formal reflux testing (n=23, 52.4% vs n=42, 46.7%, p=0.54), there was no difference in detected rates of gastroesophageal reflux disease (p=0.51). Most patients completed validated symptom questionnaires, and there were no observed differences in completion rates (71.4% vs 66.7%, p=0.58) or esophageal symptom burden between cases and controls.
Discussion: We did not observe an impact of GLP-1RA use on esophageal function assessed by HRM among patients with type 2 diabetes. These results suggest that patients may continue GLP-1RAs when undergoing esophageal testing and are at limited risk for esophageal dysmotility when using them.
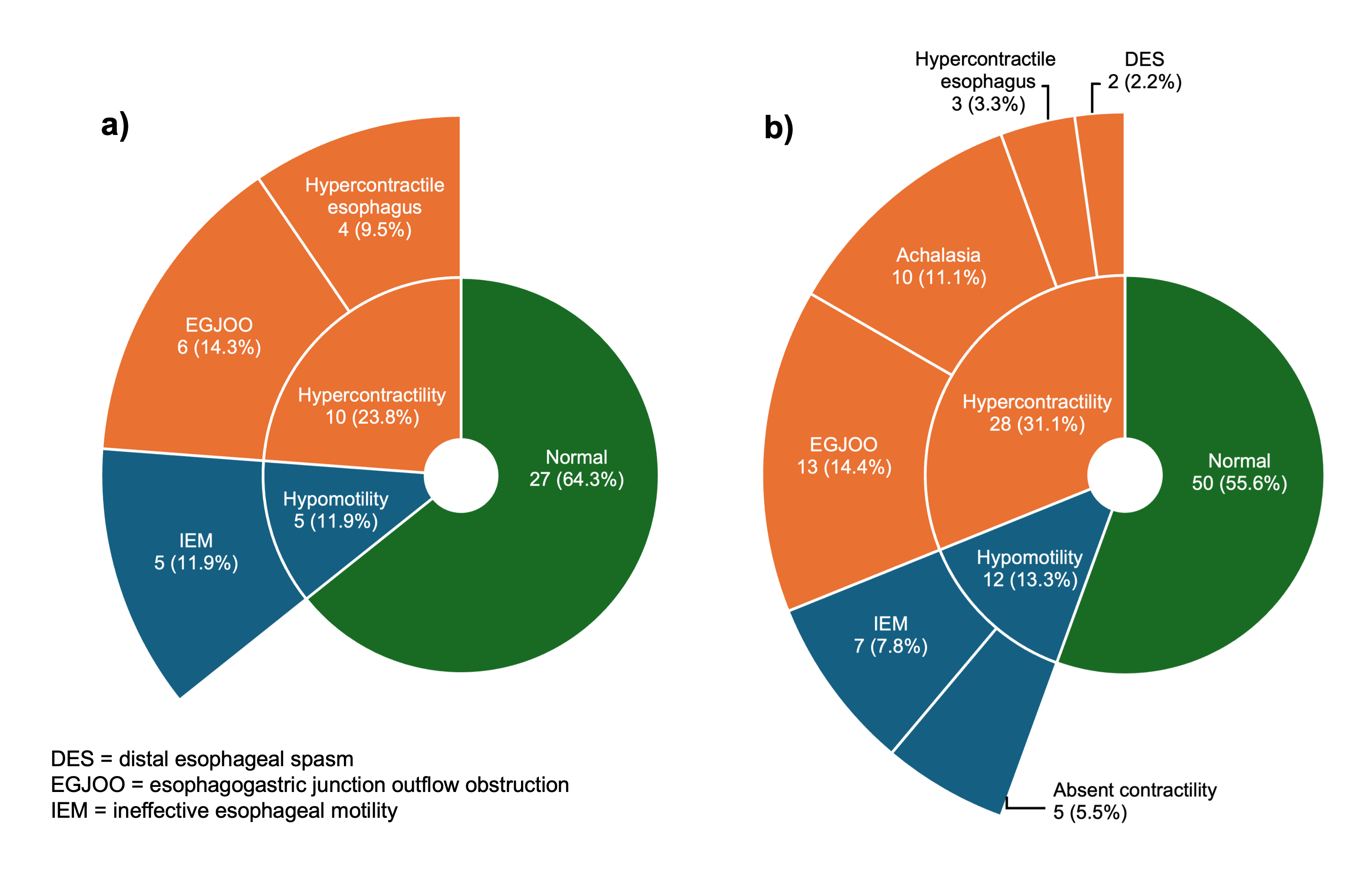
Figure: Table 1: Patient characteristics. Data are expressed as n(%) unless otherwise stated. BMI: body mass index; GERD: gastroesophageal reflux disease; GLP-1RA: glucagon-like peptide-1 receptor agonist; HRM: high-resolution manometry; IQR: interquartile range
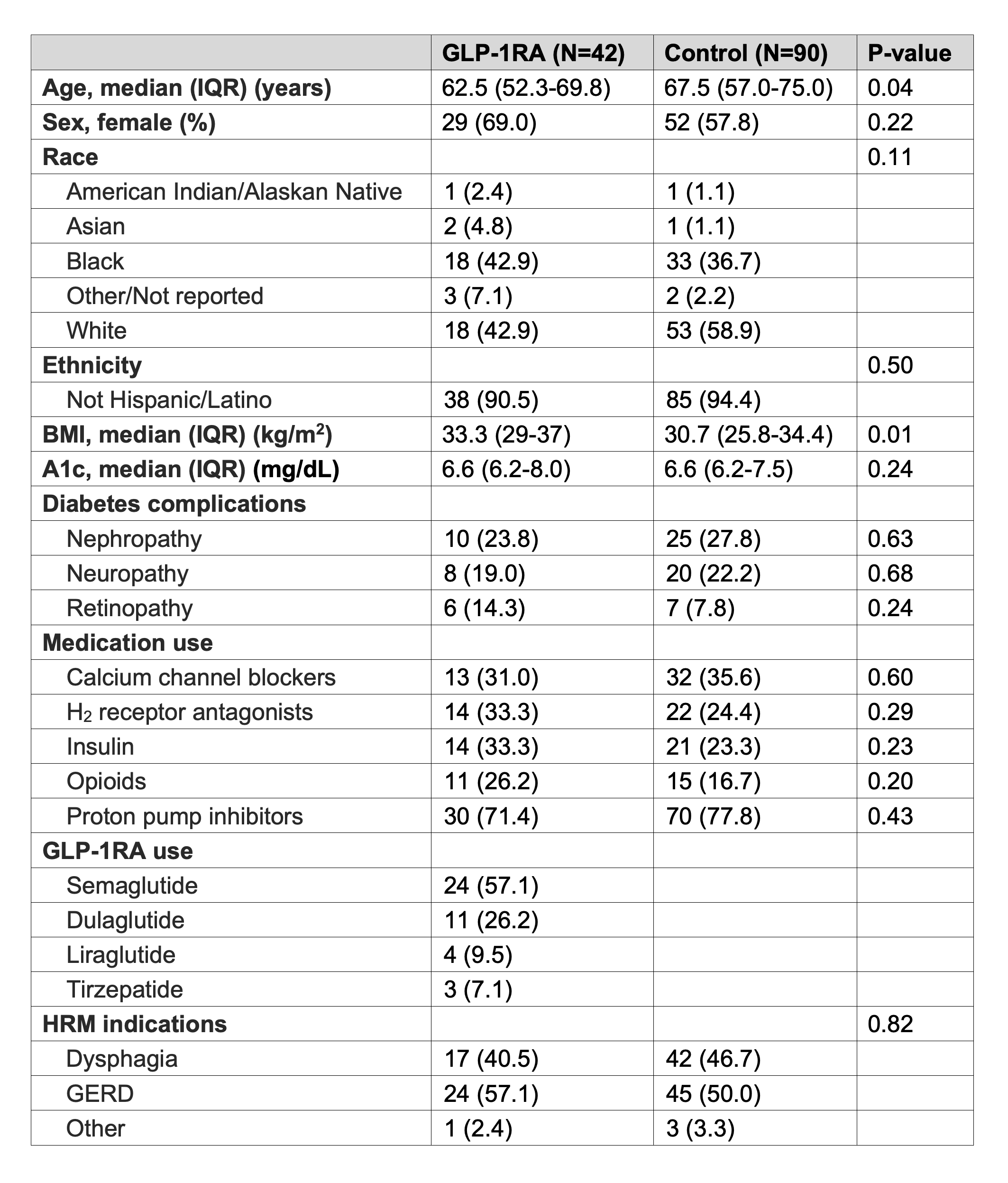
Figure: Figure 1: Esophageal motility patterns among a) GLP-1RA users and b) Controls. There were no significant differences in either individual HRM diagnoses (p=0.071) or grouped diagnoses of normal, hypomotility and hypercontractility (p=0.62) between groups. DES: distal esophageal spasm; IEM: ineffective esophageal manometry; EGJOO: esophagogastric junction outflow obstruction
Disclosures:
Annie Wang indicated no relevant financial relationships.
Laura Bach indicated no relevant financial relationships.
David Leiman: BMS – Stock-publicly held company(excluding mutual/index funds). Lilly – Consultant. Merck – Consultant. Novo Nordisk – Consultant. Regeneron – Advisor or Review Panel Member. TargetRWE – Advisory Committee/Board Member.
Annie L. Wang, BS, BA1, Laura Bach, DO2, David A. Leiman, MD, MSHP3, 61, Impact of GLP-1RA Therapy on Esophageal Motility Patterns, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
Annie L. Wang, BS, BA1, Laura Bach, DO2, David A. Leiman, MD, MSHP3
1Duke University School of Medicine, Durham, NC; 2Duke University, Durham, NC; 3Duke Clinical Research Institute, Durham, NC
Introduction: Glucagon-like peptide-1 receptor agonist (GLP-1RA) use is associated with delayed gastric emptying. It is unknown, however, whether GLP-1RAs affect esophageal motility. We aimed to assess the relationship between GLP-1RA use and esophageal function test results.
Methods: Patients with type 2 diabetes undergoing high-resolution manometry (HRM) between December 2022 and October 2024 were retrospectively evaluated, comparing cases with controls (on versus off GLP-1RA). HRM diagnoses per Chicago Classification 4.0, reflux testing results, and patient-reported symptom scores from validated questionnaires including the Brief Esophageal Disease Questionnaire, Eckardt, and Reflux Disease Questionnaire were collected. Patients with prior foregut surgery and known conditions associated with dysmotility (e.g., scleroderma) were excluded. Comparisons were performed using Chi-square and Mann-Whitney U tests for categorical and continuous measures, respectively.
Results: There were 132 patients (n=42 cases) assessed and most (n=29, 69.0%) cases were female. Compared to controls, patients receiving GLP-1RAs were younger (median 62.5 vs 67.5 years, p=0.04) and had higher BMI (median 33.3 vs 30.7 kg/m2, p=0.01), but there were no differences in type 2 diabetes control or use of acid suppression medications between groups. Semaglutide was the most commonly used GLP-1RA (n=24, 57.1%) among cases, and there was no association (p=0.51) between GLP-1RA type or dose and esophageal motility diagnoses. Most GLP-1RA users (n=27, 64.3%) had a normal HRM, and there was no significant difference (p=0.071) in overall HRM diagnoses between cases and controls. When restricting analyses to patients who underwent HRM without concomitant pH testing, there remained no significant difference in HRM diagnoses (p=0.15). Among cases and controls undergoing formal reflux testing (n=23, 52.4% vs n=42, 46.7%, p=0.54), there was no difference in detected rates of gastroesophageal reflux disease (p=0.51). Most patients completed validated symptom questionnaires, and there were no observed differences in completion rates (71.4% vs 66.7%, p=0.58) or esophageal symptom burden between cases and controls.
Discussion: We did not observe an impact of GLP-1RA use on esophageal function assessed by HRM among patients with type 2 diabetes. These results suggest that patients may continue GLP-1RAs when undergoing esophageal testing and are at limited risk for esophageal dysmotility when using them.

Figure: Table 1: Patient characteristics. Data are expressed as n(%) unless otherwise stated. BMI: body mass index; GERD: gastroesophageal reflux disease; GLP-1RA: glucagon-like peptide-1 receptor agonist; HRM: high-resolution manometry; IQR: interquartile range

Figure: Figure 1: Esophageal motility patterns among a) GLP-1RA users and b) Controls. There were no significant differences in either individual HRM diagnoses (p=0.071) or grouped diagnoses of normal, hypomotility and hypercontractility (p=0.62) between groups. DES: distal esophageal spasm; IEM: ineffective esophageal manometry; EGJOO: esophagogastric junction outflow obstruction
Disclosures:
Annie Wang indicated no relevant financial relationships.
Laura Bach indicated no relevant financial relationships.
David Leiman: BMS – Stock-publicly held company(excluding mutual/index funds). Lilly – Consultant. Merck – Consultant. Novo Nordisk – Consultant. Regeneron – Advisor or Review Panel Member. TargetRWE – Advisory Committee/Board Member.
Annie L. Wang, BS, BA1, Laura Bach, DO2, David A. Leiman, MD, MSHP3, 61, Impact of GLP-1RA Therapy on Esophageal Motility Patterns, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.



