Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
70 - Efficacy and Safety of Subcutaneous Guselkumab Rescue Therapy in Patients With Moderately to Severely Active Crohn’s Disease and Inadequate Response to Ustekinumab: Results From GALAXI 1, 2, & 3 Long-Term Extension
Wednesday, October 29, 2025
9:20 AM - 9:30 AM PDT
Location: North Ballroom 120BC

Anita Afzali, MD, MPH, MHCM, FACG
Professor of Medicine, Interim Chair, Department of Internal Medicine
University of Cincinnati College of Medicine
Cincinnati, OH
Presenting Author(s)
Anita Afzali, MD, MPH, MHCM, FACG1, Douglas Wolf, MD2, Rupert Leong, MBBS, MD, FRACP3, Rian Van Rampelbergh, MD4, Wilbert van Duijnhoven, MSc4, Christopher Busse, MS5, Tadakazu Hisamatsu, MD, PhD6, Julián Panés, MD7
1University of Cincinnati College of Medicine, Cincinnati, OH; 2Atlanta Gastroenterology Associates, Atlanta, GA; 3Macquarie University Hospital, Sydney, New South Wales, Australia; 4Johnson & Johnson, Antwerp, Antwerpen, Belgium; 5Johnson & Johnson, Horsham, PA; 6Kyorin University School of Medicine, Tokyo, Tokyo, Japan; 7Hospital Clínic de Barcelona, IDIBAPS, CIBERehd, Barcelona, Catalonia, Spain
Introduction: The phase 2b GALAXI 1 and phase 3 GALAXI 2&3 studies evaluated guselkumab(GUS), a dual-acting IL-23p19 subunit inhibitor, in pts with moderately to severely active CD. Pts treated with ustekinumab(UST) who met inadequate response criteria during the long-term extension(LTE) could switch to GUS 200mg SC q4w. We present efficacy/safety results in pts who received GUS after experiencing an inadequate response to UST in the pooled GALAXI LTE.
Methods: Individuals with prior inadequate response/intolerance to UST were excluded from GALAXI; however, during the LTE, pts treated with UST 90mg SC q8w who met inadequate treatment response criteria(not in clinical response and CDAI ≥220) between Wks52-80 were eligible for treatment switch to GUS 200mg SC q4w, without IV induction. Clinical response and clinical remission were both assessed 16wks after treatment switch. Endoscopic response and endoscopic remission were assessed at Wk96(definitions in Figure). Safety was assessed through Wk96.
Results: In total, 80 pts treated with UST underwent treatment switch to GUS 200mg SC q4w;75 pts were included in the efficacy analyses(baseline mean age, 35.2yrs; male, 64.0%; mean CD disease duration, 8.21yrs; mean CDAI, 291.5; mean SES-CD, 12.8; history of inadequate response/intolerance to biologics[BIO-IR], 60.0%).
Proportions of pts achieving clinical response and clinical remission 16wks after treatment switch from UST to GUS 200mg SC q4w were comparable to proportions of pts treated with GUS 200mg IV q4w induction in the pooled GALAXI 2&3 BIO-IR subgroup who achieved clinical response and clinical remission at Wk12(Figure).
Proportions of pts achieving endoscopic response and endoscopic remission at Wk96(ie, ~1yr after treatment switch) were similar to proportions of pts in the pooled GALAXI 2&3 BIO-IR subgroup receiving GUS 200mg SC q4w maintenance who achieved endoscopic response and endoscopic remission at Wk48.
Safety results are summarized in the Table.
Discussion: Among pts who experienced inadequate response to UST in the LTE, more than half achieved clinical remission 16wks after treatment switch to GUS 200mg SC q4w, and ~50% were in endoscopic response ~1yr after treatment switch. These data suggest pts with an inadequate treatment response to UST may benefit from GUS treatment. Results should be interpreted considering that pts received GUS SC maintenance therapy directly without IV induction. Key safety event rates were consistent with the known safety profile of GUS in approved indications.
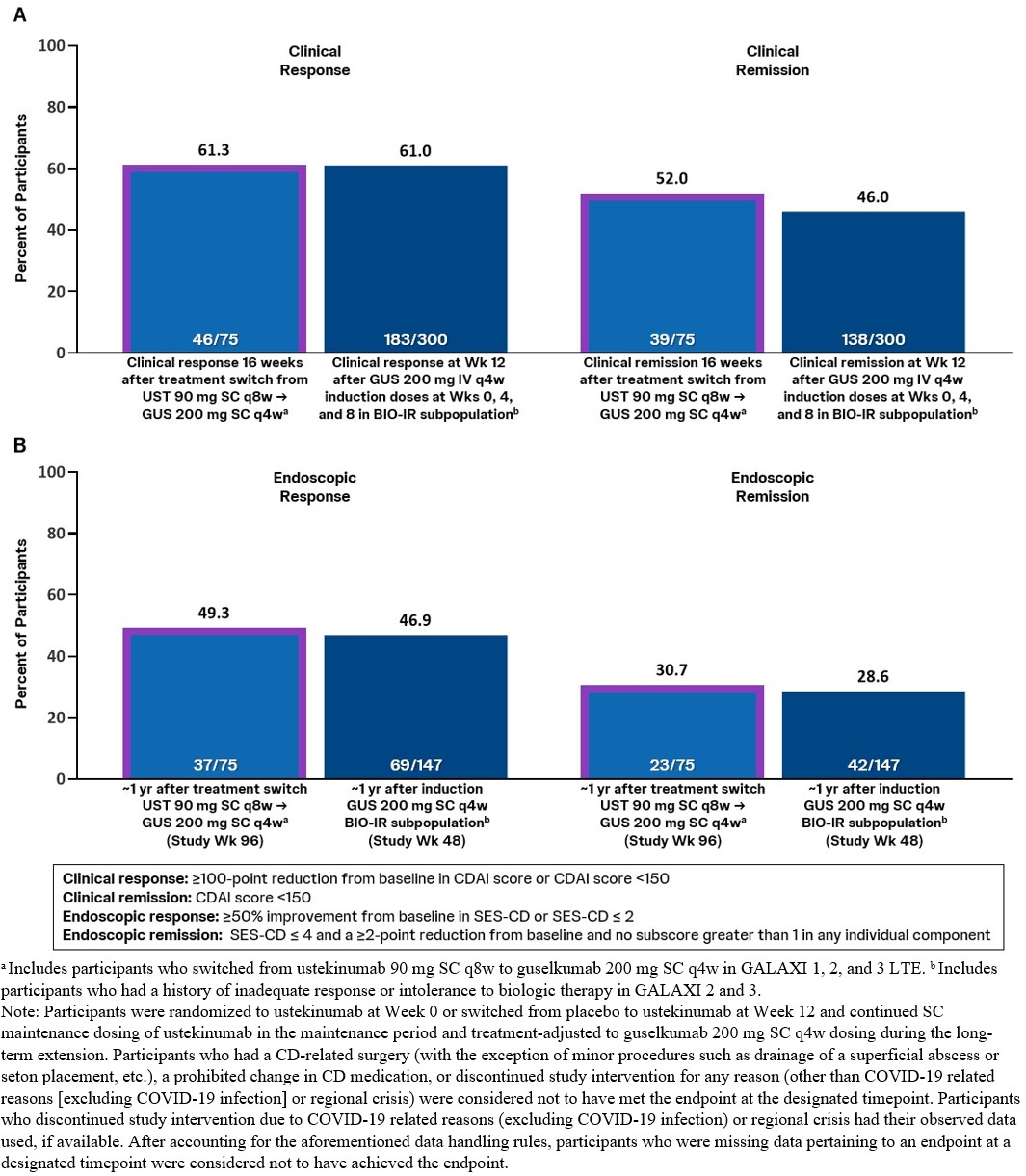
Figure: Figure. Clinical Outcomes 16 Weeks After Treatment Switch (A), and Endoscopic Outcomes at Study Week 96 (B)

Figure: Table. Safety Summary From Treatment Switch Through Week 96
Disclosures:
Anita Afzali: AbbVie – Advisory Committee/Board Member, Consultant, Speaker fees. Bristol Myers Squibb/Celgene – Consultant. DiaSorin – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Gilead – Consultant. IBD Horizons – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker fees. Pfizer – Advisory Committee/Board Member, Consultant, Speaker fees. Scrubs & Heels Foundation – Advisory Committee/Board Member, Consultant, Co-Founder. Takeda – Advisory Committee/Board Member, Consultant, Speaker fees. TLL Pharmaceuticals – Consultant.
Douglas Wolf: AbbVie – Consultant. Bristol Myers Squibb – Consultant. Janssen – Consultant. Lilly – Consultant. Pfizer – Consultant. Takeda – Consultant.
Rupert Leong: AbbVie – Advisory Committee/Board Member. Aspen – Advisory Committee/Board Member. BMS – Advisory Committee/Board Member. Celgene – Advisory Committee/Board Member. Celltrion – Advisory Committee/Board Member, Grant/Research Support. Chiesi – Advisory Committee/Board Member. Ferring – Advisory Committee/Board Member. Gastroenterological Society of Australia – Grant/Research Support. Glutagen – Advisory Committee/Board Member. Gutsy Group – Grant/Research Support. Hospira – Advisory Committee/Board Member. Janssen – Advisory Committee/Board Member, Grant/Research Support. Lilly – Advisory Committee/Board Member. McCusker Charitable Trust – Grant/Research Support. MSD – Advisory Committee/Board Member. NHMRC – Grant/Research Support. Novartis – Advisory Committee/Board Member. Pfizer – Advisory Committee/Board Member, Grant/Research Support. Prometheus Biosciences – Advisory Committee/Board Member. Shire – Grant/Research Support. Takeda – Advisory Committee/Board Member, Grant/Research Support. University of Sydney – Grant/Research Support.
Rian Van Rampelbergh: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Wilbert van Duijnhoven: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Christopher Busse: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Tadakazu Hisamatsu: AbbVie GK – Consultant, Grant/Research Support, Lecture fees. Boston Scientific Corporation – Grant/Research Support. Bristol Myers Squibb – Consultant. Daiichi Sankyo Co. Ltd. – Grant/Research Support, Honararium. EA Pharma Co. Ltd. – Consultant, Grant/Research Support, Lecture fees. Gilead Sciences – Consultant. JIMRO Co. Ltd. – Grant/Research Support, Lecture fees. Johnson & Johnson – Consultant, Lecture fees. Kissei Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Kyorin Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Lilly – Consultant. Mitsubishi Tanabe Pharma Corporation – Consultant, Grant/Research Support, Lecture fees. Mochida Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Nichi-Iko Pharmaceutical Co. Ltd – Consultant. Nippon Kayaku Co. Ltd. – Grant/Research Support. Pfizer Inc. – Consultant, Grant/Research Support, Lecture fees. Takeda Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Zeria Pharmaceutical Co – Grant/Research Support.
Julián Panés: AbbVie – Consultant. Alimentiv – Consultant, Data safety monitoring board. Athos – Consultant. Atomwise – Consultant. Boehringer Ingelheim – Consultant. Celsius – Consultant. Ferring – Consultant. Galapagos – Consultant. Genentech/Roche – Consultant. GlaxoSmithKline – Consultant. Johnson & Johnson – Consultant. Mirum – Consultant, Data safety monitoring board. Nimbus – Consultant. Pfizer – Consultant. Progenity – Consultant. Prometheus – Consultant. Protagonist – Consultant. Revolo – Consultant. Sanofi – Consultant, Data safety monitoring board. Sorriso – Consultant, Data safety monitoring board. Surrozen – Consultant, Data safety monitoring board. Takeda – Consultant. Wasserman – Consultant.
Anita Afzali, MD, MPH, MHCM, FACG1, Douglas Wolf, MD2, Rupert Leong, MBBS, MD, FRACP3, Rian Van Rampelbergh, MD4, Wilbert van Duijnhoven, MSc4, Christopher Busse, MS5, Tadakazu Hisamatsu, MD, PhD6, Julián Panés, MD7, 70, Efficacy and Safety of Subcutaneous Guselkumab Rescue Therapy in Patients With Moderately to Severely Active Crohn’s Disease and Inadequate Response to Ustekinumab: Results From GALAXI 1, 2, & 3 Long-Term Extension, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1University of Cincinnati College of Medicine, Cincinnati, OH; 2Atlanta Gastroenterology Associates, Atlanta, GA; 3Macquarie University Hospital, Sydney, New South Wales, Australia; 4Johnson & Johnson, Antwerp, Antwerpen, Belgium; 5Johnson & Johnson, Horsham, PA; 6Kyorin University School of Medicine, Tokyo, Tokyo, Japan; 7Hospital Clínic de Barcelona, IDIBAPS, CIBERehd, Barcelona, Catalonia, Spain
Introduction: The phase 2b GALAXI 1 and phase 3 GALAXI 2&3 studies evaluated guselkumab(GUS), a dual-acting IL-23p19 subunit inhibitor, in pts with moderately to severely active CD. Pts treated with ustekinumab(UST) who met inadequate response criteria during the long-term extension(LTE) could switch to GUS 200mg SC q4w. We present efficacy/safety results in pts who received GUS after experiencing an inadequate response to UST in the pooled GALAXI LTE.
Methods: Individuals with prior inadequate response/intolerance to UST were excluded from GALAXI; however, during the LTE, pts treated with UST 90mg SC q8w who met inadequate treatment response criteria(not in clinical response and CDAI ≥220) between Wks52-80 were eligible for treatment switch to GUS 200mg SC q4w, without IV induction. Clinical response and clinical remission were both assessed 16wks after treatment switch. Endoscopic response and endoscopic remission were assessed at Wk96(definitions in Figure). Safety was assessed through Wk96.
Results: In total, 80 pts treated with UST underwent treatment switch to GUS 200mg SC q4w;75 pts were included in the efficacy analyses(baseline mean age, 35.2yrs; male, 64.0%; mean CD disease duration, 8.21yrs; mean CDAI, 291.5; mean SES-CD, 12.8; history of inadequate response/intolerance to biologics[BIO-IR], 60.0%).
Proportions of pts achieving clinical response and clinical remission 16wks after treatment switch from UST to GUS 200mg SC q4w were comparable to proportions of pts treated with GUS 200mg IV q4w induction in the pooled GALAXI 2&3 BIO-IR subgroup who achieved clinical response and clinical remission at Wk12(Figure).
Proportions of pts achieving endoscopic response and endoscopic remission at Wk96(ie, ~1yr after treatment switch) were similar to proportions of pts in the pooled GALAXI 2&3 BIO-IR subgroup receiving GUS 200mg SC q4w maintenance who achieved endoscopic response and endoscopic remission at Wk48.
Safety results are summarized in the Table.
Discussion: Among pts who experienced inadequate response to UST in the LTE, more than half achieved clinical remission 16wks after treatment switch to GUS 200mg SC q4w, and ~50% were in endoscopic response ~1yr after treatment switch. These data suggest pts with an inadequate treatment response to UST may benefit from GUS treatment. Results should be interpreted considering that pts received GUS SC maintenance therapy directly without IV induction. Key safety event rates were consistent with the known safety profile of GUS in approved indications.

Figure: Figure. Clinical Outcomes 16 Weeks After Treatment Switch (A), and Endoscopic Outcomes at Study Week 96 (B)

Figure: Table. Safety Summary From Treatment Switch Through Week 96
Disclosures:
Anita Afzali: AbbVie – Advisory Committee/Board Member, Consultant, Speaker fees. Bristol Myers Squibb/Celgene – Consultant. DiaSorin – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Gilead – Consultant. IBD Horizons – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker fees. Pfizer – Advisory Committee/Board Member, Consultant, Speaker fees. Scrubs & Heels Foundation – Advisory Committee/Board Member, Consultant, Co-Founder. Takeda – Advisory Committee/Board Member, Consultant, Speaker fees. TLL Pharmaceuticals – Consultant.
Douglas Wolf: AbbVie – Consultant. Bristol Myers Squibb – Consultant. Janssen – Consultant. Lilly – Consultant. Pfizer – Consultant. Takeda – Consultant.
Rupert Leong: AbbVie – Advisory Committee/Board Member. Aspen – Advisory Committee/Board Member. BMS – Advisory Committee/Board Member. Celgene – Advisory Committee/Board Member. Celltrion – Advisory Committee/Board Member, Grant/Research Support. Chiesi – Advisory Committee/Board Member. Ferring – Advisory Committee/Board Member. Gastroenterological Society of Australia – Grant/Research Support. Glutagen – Advisory Committee/Board Member. Gutsy Group – Grant/Research Support. Hospira – Advisory Committee/Board Member. Janssen – Advisory Committee/Board Member, Grant/Research Support. Lilly – Advisory Committee/Board Member. McCusker Charitable Trust – Grant/Research Support. MSD – Advisory Committee/Board Member. NHMRC – Grant/Research Support. Novartis – Advisory Committee/Board Member. Pfizer – Advisory Committee/Board Member, Grant/Research Support. Prometheus Biosciences – Advisory Committee/Board Member. Shire – Grant/Research Support. Takeda – Advisory Committee/Board Member, Grant/Research Support. University of Sydney – Grant/Research Support.
Rian Van Rampelbergh: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Wilbert van Duijnhoven: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Christopher Busse: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Tadakazu Hisamatsu: AbbVie GK – Consultant, Grant/Research Support, Lecture fees. Boston Scientific Corporation – Grant/Research Support. Bristol Myers Squibb – Consultant. Daiichi Sankyo Co. Ltd. – Grant/Research Support, Honararium. EA Pharma Co. Ltd. – Consultant, Grant/Research Support, Lecture fees. Gilead Sciences – Consultant. JIMRO Co. Ltd. – Grant/Research Support, Lecture fees. Johnson & Johnson – Consultant, Lecture fees. Kissei Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Kyorin Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Lilly – Consultant. Mitsubishi Tanabe Pharma Corporation – Consultant, Grant/Research Support, Lecture fees. Mochida Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Nichi-Iko Pharmaceutical Co. Ltd – Consultant. Nippon Kayaku Co. Ltd. – Grant/Research Support. Pfizer Inc. – Consultant, Grant/Research Support, Lecture fees. Takeda Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Zeria Pharmaceutical Co – Grant/Research Support.
Julián Panés: AbbVie – Consultant. Alimentiv – Consultant, Data safety monitoring board. Athos – Consultant. Atomwise – Consultant. Boehringer Ingelheim – Consultant. Celsius – Consultant. Ferring – Consultant. Galapagos – Consultant. Genentech/Roche – Consultant. GlaxoSmithKline – Consultant. Johnson & Johnson – Consultant. Mirum – Consultant, Data safety monitoring board. Nimbus – Consultant. Pfizer – Consultant. Progenity – Consultant. Prometheus – Consultant. Protagonist – Consultant. Revolo – Consultant. Sanofi – Consultant, Data safety monitoring board. Sorriso – Consultant, Data safety monitoring board. Surrozen – Consultant, Data safety monitoring board. Takeda – Consultant. Wasserman – Consultant.
Anita Afzali, MD, MPH, MHCM, FACG1, Douglas Wolf, MD2, Rupert Leong, MBBS, MD, FRACP3, Rian Van Rampelbergh, MD4, Wilbert van Duijnhoven, MSc4, Christopher Busse, MS5, Tadakazu Hisamatsu, MD, PhD6, Julián Panés, MD7, 70, Efficacy and Safety of Subcutaneous Guselkumab Rescue Therapy in Patients With Moderately to Severely Active Crohn’s Disease and Inadequate Response to Ustekinumab: Results From GALAXI 1, 2, & 3 Long-Term Extension, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.

