Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4A: Endoscopy / Esophagus
60 - Impact of Sleep Positioning Device on Acid Exposure and Outcomes in Lung Transplant Recipients With Gastroesophageal Reflux Disease
Wednesday, October 29, 2025
9:20 AM - 9:30 AM PDT
Location: North Ballroom 120D
.jpg)
Scott L. Gabbard, MD, FACG
Assistant Professor
Cleveland Clinic
Cleveland, OH
Presenting Author(s)
Award: ACG Governors Award for Excellence in Clinical Research
Manik Aggarwal, MD1, Lucy Thuita, MS2, Adam Kichler, DO3, Zubin Arora, MD4, Yi L. Qin, MD5, Scott L. Gabbard, MD, FACG6
1Mayo Clinic, Rochester, MN; 2Cleveland Clinic, Cleveland, OH; 3Allegheny Health Network, Pittsburgh, PA; 4MNGI Digestive Health, Minneapolis, MN; 5Cleveland Clinic Foundation, Cleveland, OH; 6Cleveland Clinic Foundation, Shaker Heights, OH
Introduction: Gastroesophageal reflux disease (GERD) plays an important role in lung transplant (LT) outcomes. This study aims to evaluate the impact of a novel sleep positioning device (SPD) on lung function in LT recipients with GERD.
Methods: In this single center cohort study, LT recipients (2014-2019) with GERD who were prescribed an SPD 2 years of LT were included. A historical control group of LT recipients with GERD on PPI only (2011-2013) was selected to compare forced expiratory volume in 1 second (FEV1) as a marker of lung function.
Results: 20 LT recipients using SPD (cases) and 54 patients in the historical control group with proven GERD were included. Mean (standard deviation [SD]) interval between LT and SPD prescription was 8.6 (5.7) months. Mean % predicted FEV1 was higher in SPD users than controls post LT at 6mo (80% vs. 69%) and 1y (80% vs. 71%) respectively. Among SPD users, post-SPD FEV1 was higher than pre–SPD FEV1 (82% vs. 70%).
Ambulatory pH monitoring was performed pre- and post-SPD use in 15 patients. Acid exposure time in the supine position decreased significantly after SPD use when compared to pre-SPD readings (median 2.2% vs 15%, p< 0.0001); total acid exposure time also decreased significantly with SPD use (median 4.3% vs 11%, p= 0.015). Supine reflux resolved in 11 of 15 (73.1%) patients.
Discussion: Use of a novel SPD can stabilize or improve pulmonary function in patients with GERD after LT. SPD may be a safe and cost-effective adjunctive therapy for management of GERD after LT.
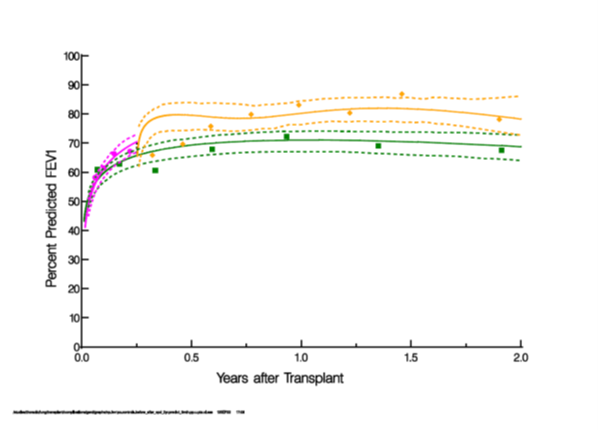
Figure: Prediction of post sleep positioning device (SPD) mean FEV1% given that SPD is prescribed at 3 months. Longitudinal model of forced expiratory volume (FEV1%) for historical controls, pre- SPD and post-SPD. Solid lines represent model-based estimates accounting for within-patient variability from repeated measurements; symbols are crude estimates that do not account for repeated measurements but are used to show resemblances of model-based estimates. Dashed lines enclose a 68% confidence band equivalent to +/- 1 standard error.
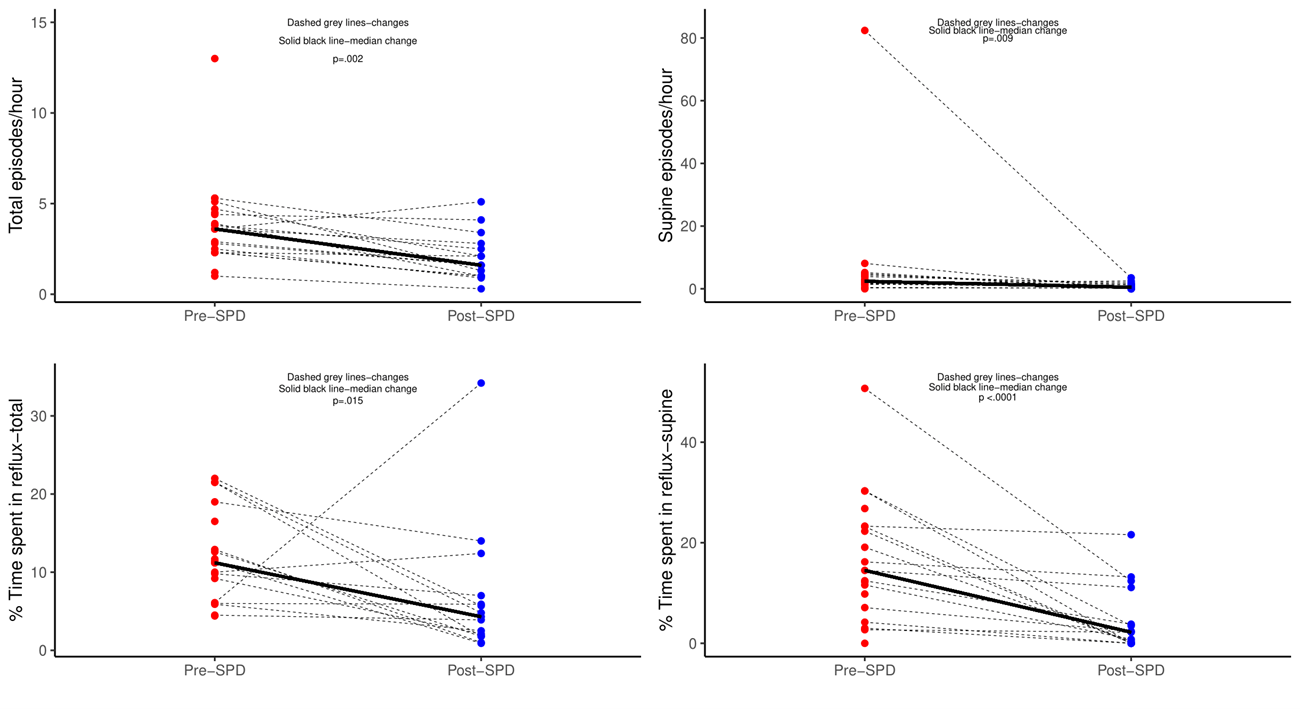
Figure: Ambulatory pH monitoring pre- and post sleep positioning device (SPD) use. The top row figures represent total (1A) and supine (1B) per hour. The bottom row figures represent changes in % time spent in reflux for total (1C) and supine (1D) episodes.
Disclosures:
Manik Aggarwal indicated no relevant financial relationships.
Lucy Thuita indicated no relevant financial relationships.
Adam Kichler indicated no relevant financial relationships.
Zubin Arora indicated no relevant financial relationships.
Yi Qin indicated no relevant financial relationships.
Scott Gabbard: Gastrogirl – invited podcast talk.
Manik Aggarwal, MD1, Lucy Thuita, MS2, Adam Kichler, DO3, Zubin Arora, MD4, Yi L. Qin, MD5, Scott L. Gabbard, MD, FACG6, 60, Impact of Sleep Positioning Device on Acid Exposure and Outcomes in Lung Transplant Recipients With Gastroesophageal Reflux Disease, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
Manik Aggarwal, MD1, Lucy Thuita, MS2, Adam Kichler, DO3, Zubin Arora, MD4, Yi L. Qin, MD5, Scott L. Gabbard, MD, FACG6
1Mayo Clinic, Rochester, MN; 2Cleveland Clinic, Cleveland, OH; 3Allegheny Health Network, Pittsburgh, PA; 4MNGI Digestive Health, Minneapolis, MN; 5Cleveland Clinic Foundation, Cleveland, OH; 6Cleveland Clinic Foundation, Shaker Heights, OH
Introduction: Gastroesophageal reflux disease (GERD) plays an important role in lung transplant (LT) outcomes. This study aims to evaluate the impact of a novel sleep positioning device (SPD) on lung function in LT recipients with GERD.
Methods: In this single center cohort study, LT recipients (2014-2019) with GERD who were prescribed an SPD 2 years of LT were included. A historical control group of LT recipients with GERD on PPI only (2011-2013) was selected to compare forced expiratory volume in 1 second (FEV1) as a marker of lung function.
Results: 20 LT recipients using SPD (cases) and 54 patients in the historical control group with proven GERD were included. Mean (standard deviation [SD]) interval between LT and SPD prescription was 8.6 (5.7) months. Mean % predicted FEV1 was higher in SPD users than controls post LT at 6mo (80% vs. 69%) and 1y (80% vs. 71%) respectively. Among SPD users, post-SPD FEV1 was higher than pre–SPD FEV1 (82% vs. 70%).
Ambulatory pH monitoring was performed pre- and post-SPD use in 15 patients. Acid exposure time in the supine position decreased significantly after SPD use when compared to pre-SPD readings (median 2.2% vs 15%, p< 0.0001); total acid exposure time also decreased significantly with SPD use (median 4.3% vs 11%, p= 0.015). Supine reflux resolved in 11 of 15 (73.1%) patients.
Discussion: Use of a novel SPD can stabilize or improve pulmonary function in patients with GERD after LT. SPD may be a safe and cost-effective adjunctive therapy for management of GERD after LT.

Figure: Prediction of post sleep positioning device (SPD) mean FEV1% given that SPD is prescribed at 3 months. Longitudinal model of forced expiratory volume (FEV1%) for historical controls, pre- SPD and post-SPD. Solid lines represent model-based estimates accounting for within-patient variability from repeated measurements; symbols are crude estimates that do not account for repeated measurements but are used to show resemblances of model-based estimates. Dashed lines enclose a 68% confidence band equivalent to +/- 1 standard error.

Figure: Ambulatory pH monitoring pre- and post sleep positioning device (SPD) use. The top row figures represent total (1A) and supine (1B) per hour. The bottom row figures represent changes in % time spent in reflux for total (1C) and supine (1D) episodes.
Disclosures:
Manik Aggarwal indicated no relevant financial relationships.
Lucy Thuita indicated no relevant financial relationships.
Adam Kichler indicated no relevant financial relationships.
Zubin Arora indicated no relevant financial relationships.
Yi Qin indicated no relevant financial relationships.
Scott Gabbard: Gastrogirl – invited podcast talk.
Manik Aggarwal, MD1, Lucy Thuita, MS2, Adam Kichler, DO3, Zubin Arora, MD4, Yi L. Qin, MD5, Scott L. Gabbard, MD, FACG6, 60, Impact of Sleep Positioning Device on Acid Exposure and Outcomes in Lung Transplant Recipients With Gastroesophageal Reflux Disease, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.



