Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
68 - Guselkumab Maintenance Dose Regimens in Patients With High Disease Activity and Severity: Subgroup Analysis of Participants With Moderately to Severely Active Crohn’s Disease in the GALAXI Phase 3 Studies
Wednesday, October 29, 2025
9:00 AM - 9:10 AM PDT
Location: North Ballroom 120BC

Anita Afzali, MD, MPH, MHCM, FACG
Professor of Medicine, Interim Chair, Department of Internal Medicine
University of Cincinnati College of Medicine
Cincinnati, OH
Presenting Author(s)
Anita Afzali, MD, MPH, MHCM, FACG1, Tadakazu Hisamatsu, MD, PhD2, David T. Rubin, MD3, Nat A. Terry, MD, PhD4, Rian Van Rampelbergh, MD5, Jacqueline Yee, MS6, Kitty Yeun Yi Wan, PhD7, Zijiang Yang, PhD4, Walter Reinisch, MD, PhD8, Bruce E. Sands, MD, MS, FACG9, Silvio Danese, MD, PhD10, Remo Panaccione, MD11
1University of Cincinnati College of Medicine, Cincinnati, OH; 2Kyorin University School of Medicine, Tokyo, Tokyo, Japan; 3University of Chicago Medicine Inflammatory Bowel Disease Center, Chicago, IL; 4Johnson & Johnson, Spring House, PA; 5Johnson & Johnson, Antwerp, Antwerpen, Belgium; 6Johnson & Johnson, Raritan, NJ; 7Johnson & Johnson, Allschwil, Schwyz, Switzerland; 8Medical University of Vienna, Spitalgasse, Wien, Austria; 9Icahn School of Medicine at Mount Sinai, New York, NY; 10IRCCS Ospedale San Raffaele and University Vita-Salute San Raffaele, Milan, Lombardia, Italy; 11University of Calgary, Calgary, AB, Canada
Introduction: Guselkumab (GUS) intravenous (IV) induction followed by subcutaneous (SC) maintenance with 200mg every 4 weeks (q4w) or 100mg every 8 weeks (q8w) was effective in the GALAXI 2 & 3 phase 3 treat-through studies of participants (pts) with moderately to severely active CD, with similar efficacy between maintenance doses in the overall study populations. Here, we evaluated the efficacy of maintenance doses in subgroups of baseline disease activity and severity and inflammatory burden at Week (Wk)12 to determine if some pts may receive additional benefit from higher dose.
Methods: Eligible pts had moderately to severely active CD (CDAI 220–450 and mean daily SF score >3 or AP score >1), SES-CD ≥6 (≥4 for isolated ileal disease), and inadequate response or intolerance to oral corticosteroids, AZA, 6-MP, MTX, or biologics. Pts were randomly assigned to GUS 200mg IV at Wk0, 4, & 8, followed by GUS 100mg SC q8w or 200mg SC q4w. The studies also included placebo and ustekinumab arms, but this analysis focused on differences between GUS maintenance doses. Wk48 endpoints included clinical remission (CDAI < 150) and endoscopic response (≥50% improvement in SES-CD or SES-CD≤2). Analyses of subgroups by baseline disease activity (CDAI >300 or SES-CD >12), and inflammatory burden (CRP >5mg/L) or endoscopic response at Wk12 were prespecified but not multiplicity controlled.
Results: In the pooled GALAXI 2 & 3 dataset, pts with high disease activity at baseline (CDAI >300 or SES-CD >12) had numerically greater clinical and endoscopic outcomes at W48 with 200mg q4w compared with 100mg q8w (Fig). Pts with baseline CDAI ≤300 or SES-CD ≤12 had similar outcomes at W48 for both maintenance doses. Pts with greater inflammatory burden (CRP >5mg/L) at W12 or who were not in endoscopic response at W12 also had numerically greater clinical and endoscopic outcomes at W48 with the 200mg dose compared with the 100mg dose (Table). Pts with lower inflammatory burden (CRP ≤5mg/L) or who were in endoscopic response at Wk12 had similar clinical and endoscopic outcomes for both maintenance doses. Results for individual GALAXI 2 & 3 studies generally trended in the same direction for all subgroups.
Discussion: Pts with high clinical or endoscopic disease severity at baseline, greater inflammatory burden after induction, or who had not achieved endoscopic response after induction showed greater clinical and endoscopic outcomes at Wk48 while receiving the 200mg SC q4w dose regimen compared with the 100mg SC q8w regimen.
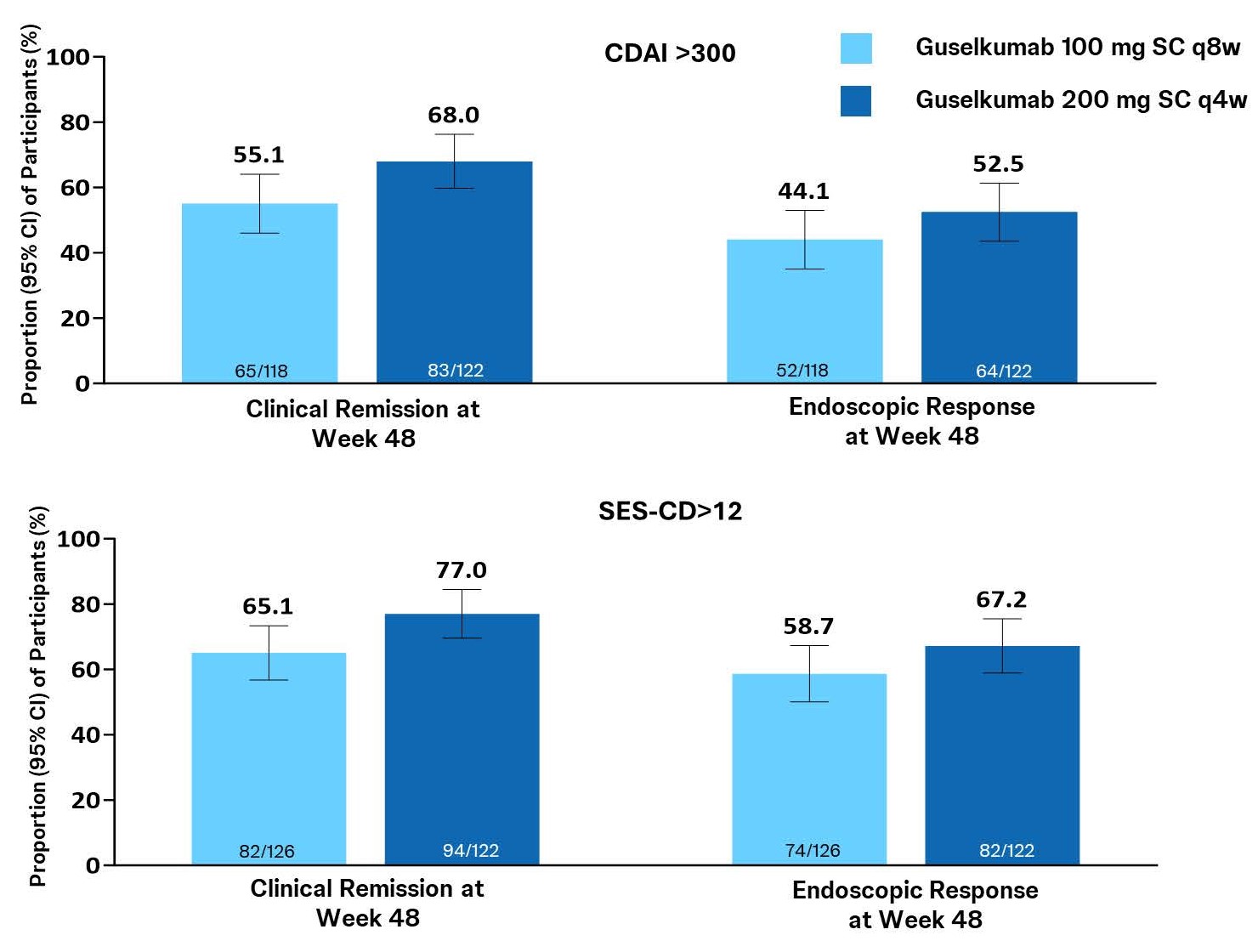
Figure: Figure. Clinal and Endoscopic Outcomes for Participants with Baseline CDAI>300 and Participants with Baseline SES-CD>12
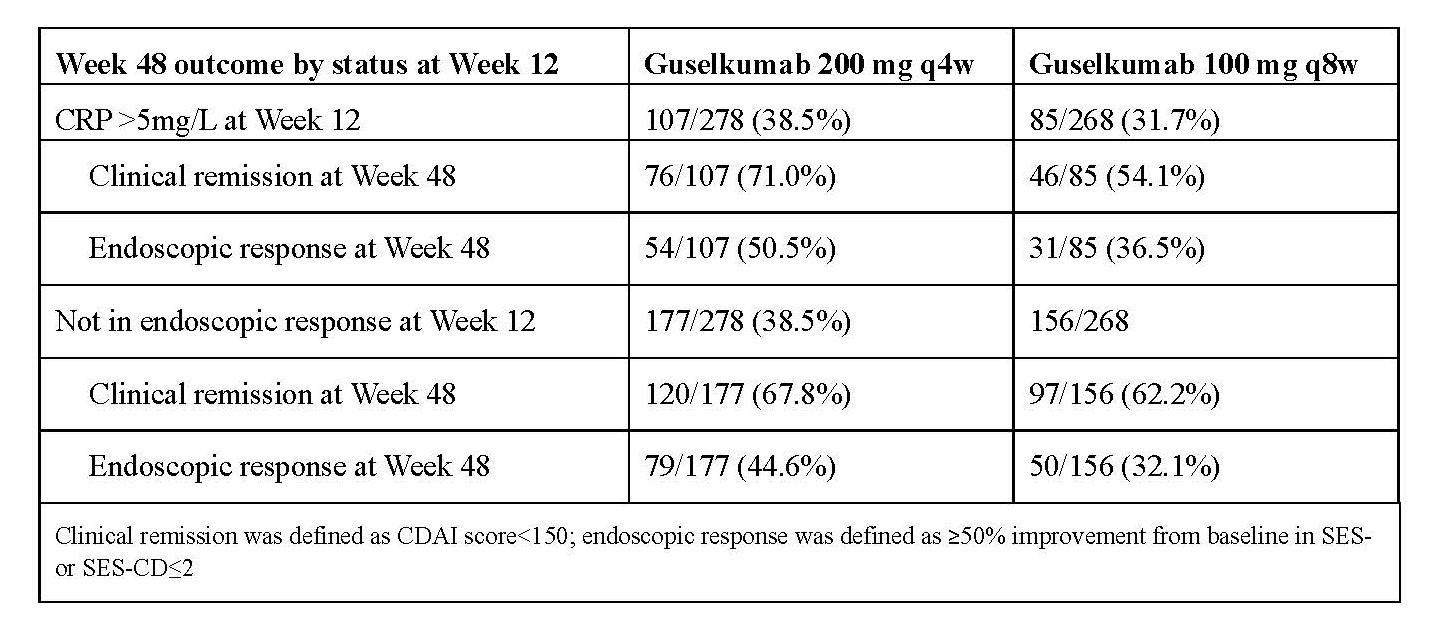
Figure: Table. Week 48 Outcomes by CRP and Endoscopic Response Status at Week 12
Disclosures:
Anita Afzali: AbbVie – Advisory Committee/Board Member, Consultant, Speaker fees. Bristol Myers Squibb/Celgene – Consultant. DiaSorin – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Gilead – Consultant. IBD Horizons – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker fees. Pfizer – Advisory Committee/Board Member, Consultant, Speaker fees. Scrubs & Heels Foundation – Advisory Committee/Board Member, Consultant, Co-Founder. Takeda – Advisory Committee/Board Member, Consultant, Speaker fees. TLL Pharmaceuticals – Consultant.
Tadakazu Hisamatsu: AbbVie GK – Consultant, Grant/Research Support, Lecture fees. Boston Scientific Corporation – Grant/Research Support. Bristol Myers Squibb – Consultant. Daiichi Sankyo Co. Ltd. – Grant/Research Support, Honararium. EA Pharma Co. Ltd. – Consultant, Grant/Research Support, Lecture fees. Gilead Sciences – Consultant. JIMRO Co. Ltd. – Grant/Research Support, Lecture fees. Johnson & Johnson – Consultant, Lecture fees. Kissei Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Kyorin Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Lilly – Consultant. Mitsubishi Tanabe Pharma Corporation – Consultant, Grant/Research Support, Lecture fees. Mochida Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Nichi-Iko Pharmaceutical Co. Ltd – Consultant. Nippon Kayaku Co. Ltd. – Grant/Research Support. Pfizer Inc. – Consultant, Grant/Research Support, Lecture fees. Takeda Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Zeria Pharmaceutical Co – Grant/Research Support.
David Rubin: AbbVie – Advisory Committee/Board Member, Consultant, Speakers Bureau. Altrubio – Advisory Committee/Board Member, Consultant, Speakers Bureau, Stock Options. Apex – Advisory Committee/Board Member, Consultant, Speakers Bureau. Avalo – Advisory Committee/Board Member, Consultant, Speakers Bureau. Bristol Myers Squibb – Advisory Committee/Board Member, Consultant, Speakers Bureau. Buhlmann Diagnostics – Advisory Committee/Board Member, Consultant, Speakers Bureau. Celgene – Advisory Committee/Board Member, Consultant, Speakers Bureau. Connect BioPharma – Advisory Committee/Board Member, Consultant, Speakers Bureau. Cornerstones Health, Inc. – Advisory Committee/Board Member. Crohn's & Colitis Foundation – Advisory Committee/Board Member. Datos Health – Stock Options. Intouch Group – Advisory Committee/Board Member, Consultant, Speakers Bureau. Iterative Health – Advisory Committee/Board Member, Consultant, Speakers Bureau, Stock Options. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speakers Bureau. Lilly – Advisory Committee/Board Member, Consultant, Speakers Bureau. Pfizer – Advisory Committee/Board Member, Consultant, Speakers Bureau. Samsung Neurologica – Advisory Committee/Board Member, Consultant, Speakers Bureau. Takeda – Advisory Committee/Board Member, Consultant, Grant/Research Support, Speakers Bureau.
Nat Terry: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Rian Van Rampelbergh: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Jacqueline Yee: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Kitty Yeun Yi Wan: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Zijiang Yang: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Walter Reinisch: AbbVie – Advisory Committee/Board Member, Consultant, Grant/Research Support. Actelion – Advisory Committee/Board Member, Consultant. Alpha Wasserman – Advisory Committee/Board Member, Consultant. AstraZeneca – Advisory Committee/Board Member, Consultant. Cellerix – Advisory Committee/Board Member, Consultant. Cosmo Pharmaceuticals – Advisory Committee/Board Member, Consultant. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Grunenthal – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant. Merck – Advisory Committee/Board Member, Consultant. Millennium – Advisory Committee/Board Member, Consultant. Novo Nordisk – Advisory Committee/Board Member, Consultant. Nycomed – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Pharmacosmos – Advisory Committee/Board Member, Consultant. Salix Pharmaceuticals – Advisory Committee/Board Member, Consultant. Schering-Plough – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB Pharma – Advisory Committee/Board Member, Consultant. Vifor Pharma – Advisory Committee/Board Member, Consultant.
Bruce Sands: AbbVie – Consultant. Abivax – Consultant, speaking fees. Adiso Therapeutics – Consultant. Agomab Therapeutics – Consultant. Alimentiv – Consultant. Amgen – Consultant. AnaptysBio – Consultant. Arena Pharmaceuticals – Consultant. Artugen Therapeutics – Consultant. Astra Zeneca – Consultant. Biolojic Design – Consultant. Biora Therapeutics – Consultant. Boehringer Ingelheim – Consultant. Boston Pharmaceuticals – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, speaking fees. Calibr – Consultant. Celgene – Consultant. Celltrion – Consultant. ClostraBio – Consultant. Eli Lilly & Company – Consultant, speaking fees. Enthera – Consultant. Enveda Biosciences – Consultant. Equillium – Consultant. Evommune – Consultant. Ferring – Consultant. Fresenius Kabi – Consultant. Fzatat – Consultant. Galapagos – Consultant. Genentech (Roche) – Consultant. Gilead Sciences – Consultant. GlaxoSmithKline – Consultant. Gossamer Bio – Consultant. Imhotex – Consultant. Index Pharmaceuticals – Consultant. Innovation Pharmaceuticals – Consultant. Inotrem – Consultant. Janssen R&D – Consultant, Grant/Research Support, speaking fees. Kaleido – Consultant. Kallyope – Consultant. Merck & Co., Inc., Rahway, NJ, USA – Consultant. Microba – Consultant. Mobius Care – Consultant. Morphic Therapeutics – Consultant. MRM Health – Consultant. Nexus Therapeutics – Consultant. Nimbus Discovery – Consultant. Odyssey Therapeutics – Consultant. Pfizer – Consultant, Grant/Research Support, speaking fees. Progenity – Consultant. Prometheus Biosciences – Consultant. Prometheus Laboratories – Consultant. Protagonist Therapeutics – Consultant. Q32 Bio – Consultant. Rasayana Therapeutics – Consultant. Recludix Therapeutics – Consultant. Reistone Biopharma – Consultant. Sanofi – Consultant. Spyre Therapeutics – Consultant. Sun Pharma – Consultant. Surrozen – Consultant. Takeda – Consultant, Grant/Research Support, speaking fees. Target RWE – Consultant. Teva – Consultant. Theravance Biopharma – Consultant, Grant/Research Support. TLL Pharmaceutical – Consultant. Tr1X – Consultant. Union Therapeutics – Consultant. Ventyx Biosciences – Consultant, Stock Options.
Silvio Danese: AbbVie – Consultant, Lecture fees. Alimentiv – Consultant. Allergan – Consultant. Amgen – Consultant, Lecture fees. AstraZeneca – Consultant. Athos Therapeutics – Consultant. Biogen – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Celltrion – Consultant. Eli Lilly – Consultant. Enthera – Consultant. F. Hoffmann-La Roche Ltd – Consultant. Ferring Pharmaceuticals Inc. – Consultant, Lecture fees. Gilead – Consultant, Lecture fees. Hospira – Consultant. Inotrem – Consultant. Johnson & Johnson – Consultant, Lecture fees. MSD – Consultant. Mundipharma – Consultant. Mylan – Consultant, Lecture fees. Pfizer – Consultant, Lecture fees. Sandoz – Consultant. Sublimity Therapeutics – Consultant. Takeda – Consultant, Lecture fees. TiGenix – Consultant. UCB Inc. – Consultant. Vifor (International) Ltd. – Consultant.
Remo Panaccione: Abbott – Consultant. AbbVie – Advisory Committee/Board Member, Consultant, Speaker's fees. Abivax – Consultant. Alimentiv – Advisory Committee/Board Member, Consultant. Amgen – Advisory Committee/Board Member, Consultant, Speaker's fees. AnaptysBio – Consultant. Arena Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. AstraZeneca – Advisory Committee/Board Member, Consultant. Biogen – Advisory Committee/Board Member, Consultant. Boehringer Ingelheim – Advisory Committee/Board Member, Consultant. Bristol Myers Squibb – Advisory Committee/Board Member, Consultant, Speaker's fees. Celgene – Advisory Committee/Board Member, Consultant, Speaker's fees. Celltrion – Consultant. Cosmos Pharmaceuticals – Consultant. Eisai – Consultant. Elan Pharmaceuticals – Consultant. Eli Lilly and Company – Advisory Committee/Board Member, Consultant, Speaker's fees. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. Fresenius Kabi – Advisory Committee/Board Member, Consultant, Speaker's fees. Galapagos – Consultant. Genentech (Roche) – Advisory Committee/Board Member, Consultant. Gilead Sciences – Advisory Committee/Board Member, Consultant, Speaker's fees. GlaxoSmithKline – Advisory Committee/Board Member, Consultant. JAMP Pharma – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker's fees. Merck & Co., Inc. – Advisory Committee/Board Member, Consultant, Speaker's fees. Mirador – Consultant. Mylan – Advisory Committee/Board Member, Consultant. Novartis – Advisory Committee/Board Member, Consultant. Odyssey – Consultant. Oppilan Pharma – Advisory Committee/Board Member, Consultant. Organon – Advisory Committee/Board Member, Consultant, Speaker's fees. Pandion Therapeutics – Advisory Committee/Board Member, Consultant. Pendopharm G.I. Solutions – Consultant. Pfizer – Advisory Committee/Board Member, Consultant, Speaker's fees. Progenity – Advisory Committee/Board Member, Consultant. Prometheus Biosciences – Consultant. Protagonist Therapeutics Inc – Advisory Committee/Board Member, Consultant. Roche – Advisory Committee/Board Member, Consultant, Speaker's fees. Sandoz – Advisory Committee/Board Member, Consultant, Speaker's fees. Sanofi – Consultant. Satisfai Health – Consultant. Shire Pharma – Advisory Committee/Board Member, Consultant, Speaker's fees. Spyre Therapeutics – Consultant. Sublimity Therapeutics – Advisory Committee/Board Member, Consultant. Takeda Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. Teva – Consultant. Theravance Biopharma – Consultant. Tillots – Consultant. Trellus Health – Consultant. UCB – Consultant. Union Biopharma – Consultant. Ventyx Biosciences – Advisory Committee/Board Member, Consultant. Viatris – Consultant.
Anita Afzali, MD, MPH, MHCM, FACG1, Tadakazu Hisamatsu, MD, PhD2, David T. Rubin, MD3, Nat A. Terry, MD, PhD4, Rian Van Rampelbergh, MD5, Jacqueline Yee, MS6, Kitty Yeun Yi Wan, PhD7, Zijiang Yang, PhD4, Walter Reinisch, MD, PhD8, Bruce E. Sands, MD, MS, FACG9, Silvio Danese, MD, PhD10, Remo Panaccione, MD11, 68, Guselkumab Maintenance Dose Regimens in Patients With High Disease Activity and Severity: Subgroup Analysis of Participants With Moderately to Severely Active Crohn’s Disease in the GALAXI Phase 3 Studies, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1University of Cincinnati College of Medicine, Cincinnati, OH; 2Kyorin University School of Medicine, Tokyo, Tokyo, Japan; 3University of Chicago Medicine Inflammatory Bowel Disease Center, Chicago, IL; 4Johnson & Johnson, Spring House, PA; 5Johnson & Johnson, Antwerp, Antwerpen, Belgium; 6Johnson & Johnson, Raritan, NJ; 7Johnson & Johnson, Allschwil, Schwyz, Switzerland; 8Medical University of Vienna, Spitalgasse, Wien, Austria; 9Icahn School of Medicine at Mount Sinai, New York, NY; 10IRCCS Ospedale San Raffaele and University Vita-Salute San Raffaele, Milan, Lombardia, Italy; 11University of Calgary, Calgary, AB, Canada
Introduction: Guselkumab (GUS) intravenous (IV) induction followed by subcutaneous (SC) maintenance with 200mg every 4 weeks (q4w) or 100mg every 8 weeks (q8w) was effective in the GALAXI 2 & 3 phase 3 treat-through studies of participants (pts) with moderately to severely active CD, with similar efficacy between maintenance doses in the overall study populations. Here, we evaluated the efficacy of maintenance doses in subgroups of baseline disease activity and severity and inflammatory burden at Week (Wk)12 to determine if some pts may receive additional benefit from higher dose.
Methods: Eligible pts had moderately to severely active CD (CDAI 220–450 and mean daily SF score >3 or AP score >1), SES-CD ≥6 (≥4 for isolated ileal disease), and inadequate response or intolerance to oral corticosteroids, AZA, 6-MP, MTX, or biologics. Pts were randomly assigned to GUS 200mg IV at Wk0, 4, & 8, followed by GUS 100mg SC q8w or 200mg SC q4w. The studies also included placebo and ustekinumab arms, but this analysis focused on differences between GUS maintenance doses. Wk48 endpoints included clinical remission (CDAI < 150) and endoscopic response (≥50% improvement in SES-CD or SES-CD≤2). Analyses of subgroups by baseline disease activity (CDAI >300 or SES-CD >12), and inflammatory burden (CRP >5mg/L) or endoscopic response at Wk12 were prespecified but not multiplicity controlled.
Results: In the pooled GALAXI 2 & 3 dataset, pts with high disease activity at baseline (CDAI >300 or SES-CD >12) had numerically greater clinical and endoscopic outcomes at W48 with 200mg q4w compared with 100mg q8w (Fig). Pts with baseline CDAI ≤300 or SES-CD ≤12 had similar outcomes at W48 for both maintenance doses. Pts with greater inflammatory burden (CRP >5mg/L) at W12 or who were not in endoscopic response at W12 also had numerically greater clinical and endoscopic outcomes at W48 with the 200mg dose compared with the 100mg dose (Table). Pts with lower inflammatory burden (CRP ≤5mg/L) or who were in endoscopic response at Wk12 had similar clinical and endoscopic outcomes for both maintenance doses. Results for individual GALAXI 2 & 3 studies generally trended in the same direction for all subgroups.
Discussion: Pts with high clinical or endoscopic disease severity at baseline, greater inflammatory burden after induction, or who had not achieved endoscopic response after induction showed greater clinical and endoscopic outcomes at Wk48 while receiving the 200mg SC q4w dose regimen compared with the 100mg SC q8w regimen.

Figure: Figure. Clinal and Endoscopic Outcomes for Participants with Baseline CDAI>300 and Participants with Baseline SES-CD>12

Figure: Table. Week 48 Outcomes by CRP and Endoscopic Response Status at Week 12
Disclosures:
Anita Afzali: AbbVie – Advisory Committee/Board Member, Consultant, Speaker fees. Bristol Myers Squibb/Celgene – Consultant. DiaSorin – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Gilead – Consultant. IBD Horizons – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker fees. Pfizer – Advisory Committee/Board Member, Consultant, Speaker fees. Scrubs & Heels Foundation – Advisory Committee/Board Member, Consultant, Co-Founder. Takeda – Advisory Committee/Board Member, Consultant, Speaker fees. TLL Pharmaceuticals – Consultant.
Tadakazu Hisamatsu: AbbVie GK – Consultant, Grant/Research Support, Lecture fees. Boston Scientific Corporation – Grant/Research Support. Bristol Myers Squibb – Consultant. Daiichi Sankyo Co. Ltd. – Grant/Research Support, Honararium. EA Pharma Co. Ltd. – Consultant, Grant/Research Support, Lecture fees. Gilead Sciences – Consultant. JIMRO Co. Ltd. – Grant/Research Support, Lecture fees. Johnson & Johnson – Consultant, Lecture fees. Kissei Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Kyorin Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Lilly – Consultant. Mitsubishi Tanabe Pharma Corporation – Consultant, Grant/Research Support, Lecture fees. Mochida Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Nichi-Iko Pharmaceutical Co. Ltd – Consultant. Nippon Kayaku Co. Ltd. – Grant/Research Support. Pfizer Inc. – Consultant, Grant/Research Support, Lecture fees. Takeda Pharmaceutical Co. Ltd. – Grant/Research Support, Lecture fees. Zeria Pharmaceutical Co – Grant/Research Support.
David Rubin: AbbVie – Advisory Committee/Board Member, Consultant, Speakers Bureau. Altrubio – Advisory Committee/Board Member, Consultant, Speakers Bureau, Stock Options. Apex – Advisory Committee/Board Member, Consultant, Speakers Bureau. Avalo – Advisory Committee/Board Member, Consultant, Speakers Bureau. Bristol Myers Squibb – Advisory Committee/Board Member, Consultant, Speakers Bureau. Buhlmann Diagnostics – Advisory Committee/Board Member, Consultant, Speakers Bureau. Celgene – Advisory Committee/Board Member, Consultant, Speakers Bureau. Connect BioPharma – Advisory Committee/Board Member, Consultant, Speakers Bureau. Cornerstones Health, Inc. – Advisory Committee/Board Member. Crohn's & Colitis Foundation – Advisory Committee/Board Member. Datos Health – Stock Options. Intouch Group – Advisory Committee/Board Member, Consultant, Speakers Bureau. Iterative Health – Advisory Committee/Board Member, Consultant, Speakers Bureau, Stock Options. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speakers Bureau. Lilly – Advisory Committee/Board Member, Consultant, Speakers Bureau. Pfizer – Advisory Committee/Board Member, Consultant, Speakers Bureau. Samsung Neurologica – Advisory Committee/Board Member, Consultant, Speakers Bureau. Takeda – Advisory Committee/Board Member, Consultant, Grant/Research Support, Speakers Bureau.
Nat Terry: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Rian Van Rampelbergh: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Jacqueline Yee: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Kitty Yeun Yi Wan: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Zijiang Yang: Johnson & Johnson – Employee, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Walter Reinisch: AbbVie – Advisory Committee/Board Member, Consultant, Grant/Research Support. Actelion – Advisory Committee/Board Member, Consultant. Alpha Wasserman – Advisory Committee/Board Member, Consultant. AstraZeneca – Advisory Committee/Board Member, Consultant. Cellerix – Advisory Committee/Board Member, Consultant. Cosmo Pharmaceuticals – Advisory Committee/Board Member, Consultant. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Grunenthal – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant. Merck – Advisory Committee/Board Member, Consultant. Millennium – Advisory Committee/Board Member, Consultant. Novo Nordisk – Advisory Committee/Board Member, Consultant. Nycomed – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Pharmacosmos – Advisory Committee/Board Member, Consultant. Salix Pharmaceuticals – Advisory Committee/Board Member, Consultant. Schering-Plough – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB Pharma – Advisory Committee/Board Member, Consultant. Vifor Pharma – Advisory Committee/Board Member, Consultant.
Bruce Sands: AbbVie – Consultant. Abivax – Consultant, speaking fees. Adiso Therapeutics – Consultant. Agomab Therapeutics – Consultant. Alimentiv – Consultant. Amgen – Consultant. AnaptysBio – Consultant. Arena Pharmaceuticals – Consultant. Artugen Therapeutics – Consultant. Astra Zeneca – Consultant. Biolojic Design – Consultant. Biora Therapeutics – Consultant. Boehringer Ingelheim – Consultant. Boston Pharmaceuticals – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, speaking fees. Calibr – Consultant. Celgene – Consultant. Celltrion – Consultant. ClostraBio – Consultant. Eli Lilly & Company – Consultant, speaking fees. Enthera – Consultant. Enveda Biosciences – Consultant. Equillium – Consultant. Evommune – Consultant. Ferring – Consultant. Fresenius Kabi – Consultant. Fzatat – Consultant. Galapagos – Consultant. Genentech (Roche) – Consultant. Gilead Sciences – Consultant. GlaxoSmithKline – Consultant. Gossamer Bio – Consultant. Imhotex – Consultant. Index Pharmaceuticals – Consultant. Innovation Pharmaceuticals – Consultant. Inotrem – Consultant. Janssen R&D – Consultant, Grant/Research Support, speaking fees. Kaleido – Consultant. Kallyope – Consultant. Merck & Co., Inc., Rahway, NJ, USA – Consultant. Microba – Consultant. Mobius Care – Consultant. Morphic Therapeutics – Consultant. MRM Health – Consultant. Nexus Therapeutics – Consultant. Nimbus Discovery – Consultant. Odyssey Therapeutics – Consultant. Pfizer – Consultant, Grant/Research Support, speaking fees. Progenity – Consultant. Prometheus Biosciences – Consultant. Prometheus Laboratories – Consultant. Protagonist Therapeutics – Consultant. Q32 Bio – Consultant. Rasayana Therapeutics – Consultant. Recludix Therapeutics – Consultant. Reistone Biopharma – Consultant. Sanofi – Consultant. Spyre Therapeutics – Consultant. Sun Pharma – Consultant. Surrozen – Consultant. Takeda – Consultant, Grant/Research Support, speaking fees. Target RWE – Consultant. Teva – Consultant. Theravance Biopharma – Consultant, Grant/Research Support. TLL Pharmaceutical – Consultant. Tr1X – Consultant. Union Therapeutics – Consultant. Ventyx Biosciences – Consultant, Stock Options.
Silvio Danese: AbbVie – Consultant, Lecture fees. Alimentiv – Consultant. Allergan – Consultant. Amgen – Consultant, Lecture fees. AstraZeneca – Consultant. Athos Therapeutics – Consultant. Biogen – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Celltrion – Consultant. Eli Lilly – Consultant. Enthera – Consultant. F. Hoffmann-La Roche Ltd – Consultant. Ferring Pharmaceuticals Inc. – Consultant, Lecture fees. Gilead – Consultant, Lecture fees. Hospira – Consultant. Inotrem – Consultant. Johnson & Johnson – Consultant, Lecture fees. MSD – Consultant. Mundipharma – Consultant. Mylan – Consultant, Lecture fees. Pfizer – Consultant, Lecture fees. Sandoz – Consultant. Sublimity Therapeutics – Consultant. Takeda – Consultant, Lecture fees. TiGenix – Consultant. UCB Inc. – Consultant. Vifor (International) Ltd. – Consultant.
Remo Panaccione: Abbott – Consultant. AbbVie – Advisory Committee/Board Member, Consultant, Speaker's fees. Abivax – Consultant. Alimentiv – Advisory Committee/Board Member, Consultant. Amgen – Advisory Committee/Board Member, Consultant, Speaker's fees. AnaptysBio – Consultant. Arena Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. AstraZeneca – Advisory Committee/Board Member, Consultant. Biogen – Advisory Committee/Board Member, Consultant. Boehringer Ingelheim – Advisory Committee/Board Member, Consultant. Bristol Myers Squibb – Advisory Committee/Board Member, Consultant, Speaker's fees. Celgene – Advisory Committee/Board Member, Consultant, Speaker's fees. Celltrion – Consultant. Cosmos Pharmaceuticals – Consultant. Eisai – Consultant. Elan Pharmaceuticals – Consultant. Eli Lilly and Company – Advisory Committee/Board Member, Consultant, Speaker's fees. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. Fresenius Kabi – Advisory Committee/Board Member, Consultant, Speaker's fees. Galapagos – Consultant. Genentech (Roche) – Advisory Committee/Board Member, Consultant. Gilead Sciences – Advisory Committee/Board Member, Consultant, Speaker's fees. GlaxoSmithKline – Advisory Committee/Board Member, Consultant. JAMP Pharma – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant, Speaker's fees. Merck & Co., Inc. – Advisory Committee/Board Member, Consultant, Speaker's fees. Mirador – Consultant. Mylan – Advisory Committee/Board Member, Consultant. Novartis – Advisory Committee/Board Member, Consultant. Odyssey – Consultant. Oppilan Pharma – Advisory Committee/Board Member, Consultant. Organon – Advisory Committee/Board Member, Consultant, Speaker's fees. Pandion Therapeutics – Advisory Committee/Board Member, Consultant. Pendopharm G.I. Solutions – Consultant. Pfizer – Advisory Committee/Board Member, Consultant, Speaker's fees. Progenity – Advisory Committee/Board Member, Consultant. Prometheus Biosciences – Consultant. Protagonist Therapeutics Inc – Advisory Committee/Board Member, Consultant. Roche – Advisory Committee/Board Member, Consultant, Speaker's fees. Sandoz – Advisory Committee/Board Member, Consultant, Speaker's fees. Sanofi – Consultant. Satisfai Health – Consultant. Shire Pharma – Advisory Committee/Board Member, Consultant, Speaker's fees. Spyre Therapeutics – Consultant. Sublimity Therapeutics – Advisory Committee/Board Member, Consultant. Takeda Pharmaceuticals – Advisory Committee/Board Member, Consultant, Speaker's fees. Teva – Consultant. Theravance Biopharma – Consultant. Tillots – Consultant. Trellus Health – Consultant. UCB – Consultant. Union Biopharma – Consultant. Ventyx Biosciences – Advisory Committee/Board Member, Consultant. Viatris – Consultant.
Anita Afzali, MD, MPH, MHCM, FACG1, Tadakazu Hisamatsu, MD, PhD2, David T. Rubin, MD3, Nat A. Terry, MD, PhD4, Rian Van Rampelbergh, MD5, Jacqueline Yee, MS6, Kitty Yeun Yi Wan, PhD7, Zijiang Yang, PhD4, Walter Reinisch, MD, PhD8, Bruce E. Sands, MD, MS, FACG9, Silvio Danese, MD, PhD10, Remo Panaccione, MD11, 68, Guselkumab Maintenance Dose Regimens in Patients With High Disease Activity and Severity: Subgroup Analysis of Participants With Moderately to Severely Active Crohn’s Disease in the GALAXI Phase 3 Studies, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.

