Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
65 - Efficacy and Safety of Icotrokinra, a Targeted Oral Peptide That Selectively Blocks IL-23 Receptor Activation, in Ulcerative Colitis: Results From Week 28 of ANTHEM-UC, a Phase 2b Dose-Ranging Trial
Wednesday, October 29, 2025
8:30 AM - 8:40 AM PDT
Location: North Ballroom 120BC

Vipul Jairath, MBChB, DPhil, MRCP
Department of Medicine and Department of Epidemiology and Biostatistics, Western University
London, ON, Canada
Presenting Author(s)
Vipul Jairath, MBChB, DPhil, MRCP1, Britta Siegmund, MD2, Lindsey Surace, MD3, Ngozi Erondu, MD, PhD3, Minhu Chen, MD, PhD4, Karen Chachu, MD, PhD5, Edouard Louis, MD, PhD6, Katsuyoshi Matsuoka, MD, PhD7, Jimmy Limdi, MD8, Edmund Arthur, PharmD, MBA3, Nicole Houck, BS3, Mary Ellen Frustaci, BS, MS3, Joyce Zhan, PhD3, Grazyna Rydzewska, MD, PhD9, Edward V.. Loftus, MD10, Maria T.. Abreu, MD11
1Western University, London, ON, Canada; 2Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Berlin, Germany; 3Johnson & Johnson, Spring House, PA; 4The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China; 5Duke University School of Medicine, Durham, NC; 6CHU Liège University Hospital, Liège, Liege, Belgium; 7Toho University Sakura Medical Center, Chiba, Chiba, Japan; 8Northern Care Alliance NHS Foundation Trust and University of Manchester, Manchester, England, United Kingdom; 9National Medical Institute of Ministry of Interior and Administration, Warsaw, Mazowieckie, Poland; 10Mayo Clinic College of Medicine and Science and Mayo Clinic, Rochester, MN; 11Cedars-Sinai, Los Angeles, CA
Introduction: As previously reported, all 3 doses of icotrokinra (ICO; JNJ-2113) — a first-in-class targeted oral peptide that selectively blocks IL-23 receptor activation — met the week (W) 12 primary endpoint in ANTHEM-UC, a Phase 2b, randomized, double-blind, placebo-controlled, treat-through, dose-ranging study in adults with moderate to severe ulcerative colitis (UC). Here we report W28 efficacy and safety of ICO from ANTHEM-UC.
Methods: Participants (pts) had a modified Mayo score (mMS) of 5–9 inclusive, a Mayo endoscopy subscore (MES) ≥2, and inadequate response/intolerance (IR) to TNFα blockers, vedolizumab, ustekinumab, JAK inhibitors, or S1P receptor modulators (BIO/JAKi/S1P-IR) or IR to corticosteroids, AZA, or 6-MP. Randomization (1:1:1:1) to once-daily (qd) oral ICO 100 mg, 200 mg, 400 mg or placebo (PBO) was stratified by BIO/JAKi/S1P-IR status (Y/N) and MES (2 or 3).
Clinical response at W12 was the primary endpoint. Clinical response, clinical remission, symptomatic remission, endoscopic improvement, and histologic-endoscopic mucosal improvement (HEMI) were further evaluated at W28 as exploratory endpoints. Pts meeting inadequate response criteria at W16 had a treatment adjustment: PBO pts switched to ICO 400 mg qd and ICO pts had a sham adjustment. All pts meeting inadequate response criteria were considered non-responders moving forward.
Results: Analyses included all 252 randomized pts who received study medication: mean mMS, 6.63; mMS >7, 31.9%; MES=3, 58.7%; BIO/JAKi/S1P-IR, 43.3%. W16 inadequate response criteria were met by 24 (38.1%), 11 (17.2%), 11 (17.7%) and 7 (11.1%) pts receiving PBO & ICO 100, 200 and 400 mg.
Relative to PBO, all ICO doses demonstrated clinically meaningful rates of clinical response, clinical remission, symptomatic remission, endoscopic improvement, and HEMI at W28 (Figure). Relative to W12 outcomes, rates of clinical response, clinical remission, endoscopic improvement, and HEMI continued to increase through W28 in each ICO dose group.
Through W28, ≥1 AE (61.9%, 65.6%, 66.1%, 60.3%), SAEs (9.5%, 0%, 4.8%, 1.6%), or AEs leading to discontinuation of study agent (11.1%, 0%, 6.5%, 3.2%) were reported in pts in the PBO & ICO 100, 200 and 400 mg groups. No serious or opportunistic infections, tuberculosis, malignancies, clinically important hepatic disorders, VTE, MACE, or deaths were reported with ICO.
Discussion: In ANTHEM-UC, clinical efficacy and a favorable safety profile were observed with once-daily ICO through W28 in pts with moderate to severe UC.
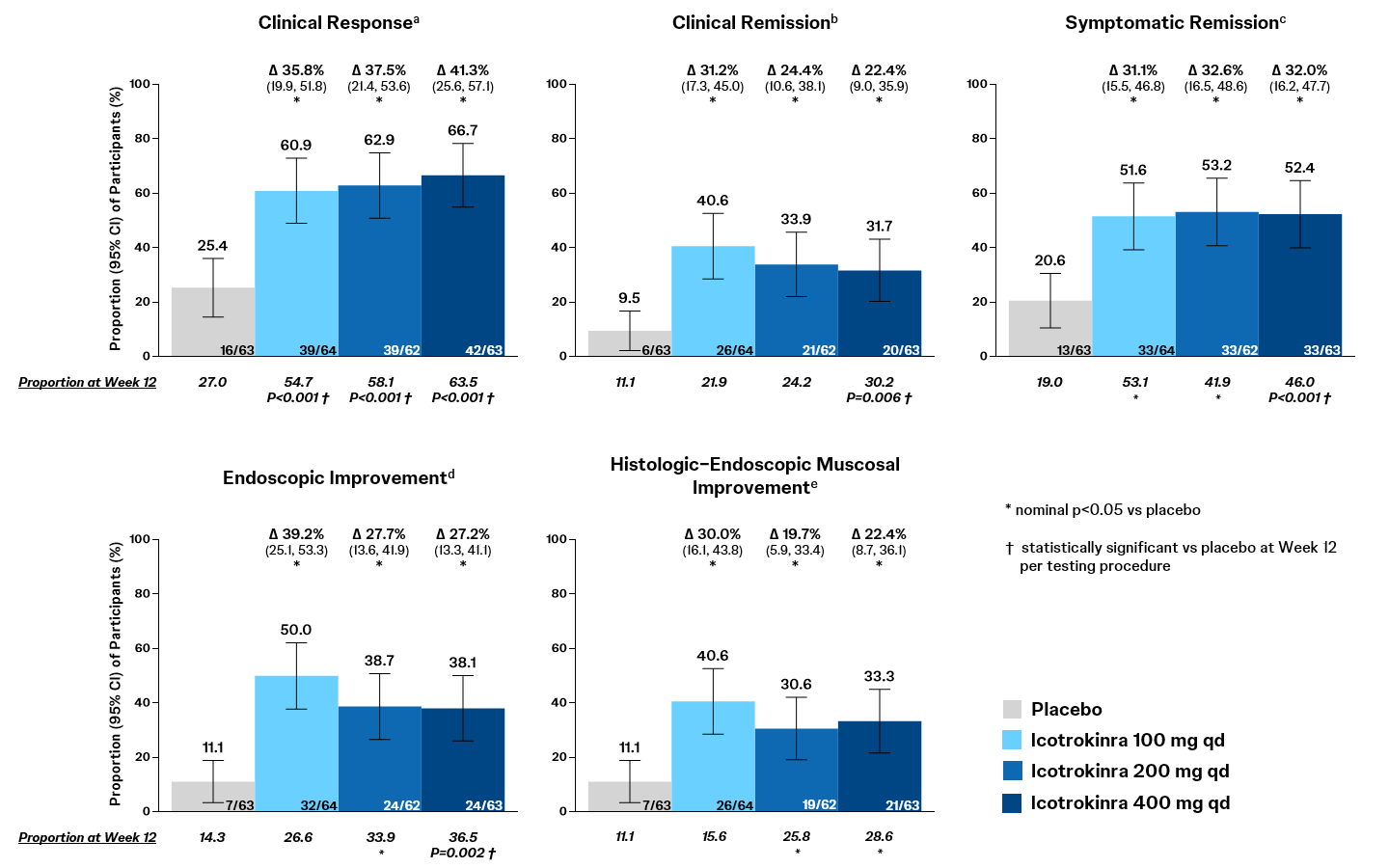
Figure: Week 28 Outcomes in ANTHEM-UC:
Data presented as percentage of participants attaining the endpoint, with adjusted treatment difference (Δ), 95% confidence interval (CI) for the adjusted treatment difference, and p-value versus placebo. At Week 28, all endpoints are exploratory and p-values are nominal. Outcomes at Week 12 that reached statistical significance per the prespecified testing procedure are indicated.
Adjusted treatment differences, 95% CIs, and p-values were based on the common risk difference by use of Mantel-Haenszel stratum weights and the Sato variance estimator, using stratification factors of inadequate response/intolerance to TNF blockers, vedolizumab, ustekinumab, JAK inhibitors, or S1P receptor modulators (Yes or No) and Mayo endoscopy subscore (MES) (2 [moderate] or 3 [severe]).
Participants with intercurrent events of ostomy or colectomy, prohibited changes in UC medication, discontinuation of study intervention for any reason except those due to major disruptions (e.g., COVID-19 related reasons or regional crisis, excluding COVID-19 infection), or meeting Week 16 inadequate response criteria were considered nonresponders for the Week 28 endpoints, as were participants who had missing data for the endpoint.
ENDPOINT DEFINITIONS: a. Clinical response: a decrease from baseline in modified Mayo score by ≥30% and ≥2 points with either a ≥1-point decrease from baseline in rectal bleeding subscore or a rectal bleeding subscore of 0 or 1. b. Clinical remission: stool frequency subscore of 0 or 1, rectal bleeding subscore of 0, and MES of 0 or 1. c. Symptomatic remission: stool frequency subscore of 0 or 1 and rectal bleeding subscore of 0
d. Endoscopic improvement: MES of 0 or 1. e. Histologic-endoscopic mucosal improvement (HEMI): histologic remission (absence of neutrophils from the mucosa [both lamina propria and epithelium], no crypt destruction, and no erosions, ulcerations, or granulation tissue according to the Geboes grading system) AND endoscopic improvement (MES of 0 or 1).
Disclosures:
Vipul Jairath: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Alimentiv – Consultant, Grant/Research Support. Arena Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Asahi Kasei Pharma – Consultant, Grant/Research Support, Speakers Bureau. Asieris Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. AstraZeneca – Consultant, Grant/Research Support, Speakers Bureau. Avoro Capital – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, Speakers Bureau. Celltrion – Consultant, Grant/Research Support, Speakers Bureau. Eli Lilly and Company – Consultant, Grant/Research Support, Speakers Bureau. Endpoint Health – Consultant. Enthera – Consultant. Ferring Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Flagship Pioneering – Consultant, Grant/Research Support, Speakers Bureau. Fresenius Kabi – Consultant, Grant/Research Support, Speakers Bureau. Galapagos NV – Consultant, Grant/Research Support, Speakers Bureau. Genentech – Consultant, Grant/Research Support, Speakers Bureau. Gilde Healthcare – Consultant. Gilead Sciences – Consultant, Grant/Research Support, Speakers Bureau. GlaxoSmithKline – Consultant, Grant/Research Support, Speakers Bureau. Innomar – Consultant. JAMP – Consultant. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Merck – Consultant, Grant/Research Support, Speakers Bureau. Metacrine – Consultant, Grant/Research Support, Speakers Bureau. Mylan – Consultant, Grant/Research Support, Speakers Bureau. Pandion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Pendopharm – Consultant, Grant/Research Support, Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. Prometheus Therapeutics and Diagnostics – Consultant, Grant/Research Support, Speakers Bureau. Protagonist Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Reistone Biopharma – Consultant, Grant/Research Support, Speakers Bureau. Roche – Consultant, Grant/Research Support, Speakers Bureau. Roivant – Consultant. Sandoz – Consultant, Grant/Research Support, Speakers Bureau. Second Genome – Consultant, Grant/Research Support, Speakers Bureau. Shire – Speakers Bureau. Sorriso Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Syndegen – Consultant. Takeda – Consultant, Intellectual Property/Patents, Speakers Bureau. TD Securities – Consultant. Teva – Consultant, Grant/Research Support, Speakers Bureau. Topivert – Consultant, Grant/Research Support, Speakers Bureau. Ventyx Biosciences – Consultant, Grant/Research Support, Speakers Bureau. Vividion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau.
Britta Siegmund: AbbVie – Consultant, Speakers Bureau. Abivax – Consultant. AlfaSigma – Speakers Bureau. Boehringer Ingelheim – Consultant. Bristol Myers Squibb – Consultant, Speakers Bureau. CED Service GmbH – Speakers Bureau. Dr. Falk Pharma – Consultant, Speakers Bureau. Eli Lilly – Consultant, Speakers Bureau. Endpoint Health – Consultant. Ferring – Speakers Bureau. Galapagos – Consultant, Speakers Bureau. Gilead – Consultant. Johnson & Johnson – Consultant, Speakers Bureau. Landos – Consultant. Materia Prima – Consultant. MSD – Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. PredictImmune – Consultant. Takeda – Consultant, Speakers Bureau. Tr1X – Speakers Bureau.
Lindsey Surace: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Ngozi Erondu: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Minhu Chen: AbbVie – Provided Educational Activities. Boehringer Ingelheim – Advisory Committee/Board Member. China Medical System – Provided Educational Activities. IPSEN – Provided Educational Activities. Johnson & Johnson – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Takeda – Grant/Research Support, Provided Educational Activities.
Karen Chachu: Johnson & Johnson – Advisor or Review Panel Member. OptumRx – Consultant.
Edouard Louis: AbbVie – Advisor or Review Panel Member, Consultant, Grant/Research Support, Speakers Bureau. Aboleris – Consultant. Biokuris – Consultant. BMS – Advisor or Review Panel Member, Speakers Bureau. Celgene – Advisory Committee/Board Member. Celltrion – Grant/Research Support, Speakers Bureau. Eli Lilly – Advisory Committee/Board Member. Falk – Grant/Research Support. Ferring – Advisor or Review Panel Member, Speakers Bureau. Fresenius-Kabi – Grant/Research Support, Speakers Bureau. Galapagos – Speakers Bureau. Johnson & Johnson – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Pfizer – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Takeda – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Thabor – Consultant.
Katsuyoshi Matsuoka: AbbVie – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Bristol Myers Squibb – Advisor or Review Panel Member. Celltrion Healthcare – Advisor or Review Panel Member. EA Pharma – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Eli Lilly – Advisor or Review Panel Member, Speakers Bureau. Gilead – Advisor or Review Panel Member, Speakers Bureau. JIMRO – Grant/Research Support. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Kyorin – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Mitsubishi Tanabe Pharma – Grant/Research Support, Speakers Bureau. Mochida – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Nippon Kayaku – Grant/Research Support, Speakers Bureau. Takeda – Advisor or Review Panel Member, Speakers Bureau. Zeria – Grant/Research Support, Speakers Bureau.
Jimmy Limdi: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Abivax – Consultant, Speakers Bureau. AlfaSigma – Consultant, Speakers Bureau. Biohit – Consultant, Speakers Bureau. Bristol Myers Squibb – Consultant, Speakers Bureau. Celltrion – Consultant, Speakers Bureau. Eli Lilly and Company – Consultant, Speakers Bureau. Ferring – Consultant, Speakers Bureau. Galapagos – Consultant, Grant/Research Support, Speakers Bureau. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. MSD – Consultant, Speakers Bureau. Pfizer – Consultant, Speakers Bureau. Takeda – Consultant, Grant/Research Support, Speakers Bureau.
Edmund Arthur: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Nicole Houck: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Mary Ellen Frustaci: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Joyce Zhan: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Grazyna Rydzewska: AbbVie – Consultant, travel/accommodation meeting expenses, Speakers Bureau. AlfaSigma – Consultant, Speakers Bureau. AstraZeneca – Consultant, Speakers Bureau. Bayer – Consultant, Speakers Bureau. Biocodex – Consultant, Speakers Bureau. Bristol Myers Squibb – Consultant, Speakers Bureau. Ferring – Consultant, travel/accommodation meeting expenses, Speakers Bureau. Johnson & Johnson – Consultant, Speakers Bureau. Lilly – Consultant, Speakers Bureau. PRO.MED.Pl – Consultant, Speakers Bureau. Recordati – Consultant, Speakers Bureau. Sanprobi – Consultant, Speakers Bureau. SOBI – Consultant, Speakers Bureau. Takeda – Consultant, travel/accommodation meeting expenses, Speakers Bureau.
Edward Loftus: AbbVie – Consultant, Grant/Research Support. Abivax – Consultant. Astellas – Consultant. Avalo – Consultant. Biocon – Consultant. Celltrion – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Exact Sciences – Stock Options. Fresenius Kabi – Consultant. Genentech – Advisory Committee/Board Member. Gilead – Consultant, Grant/Research Support. Iota Biosciences – Consultant. Iterative Health – Consultant. Johnson & Johnson – Consultant, Grant/Research Support. Merck – Consultant, Grant/Research Support. Moderna – Stock Options. Morphic – Consultant. Ono Pharma – Consultant. Spyre – Advisory Committee/Board Member. Takeda – Consultant, Grant/Research Support. TR1X Bio – Consultant.
Maria Abreu: AbbVie – Consultant. Alimentiv – Lecture fees. Arena Pharmaceuticals – Consultant. Bristol Myers Squibb – Consultant. Celsius Therapeutics – Consultant. Gilead – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Lecture fees. Lilly – Consultant. Pfizer – Consultant. Prime – Lecture fees. Prometheus Biosciences – Consultant. UCB – Consultant. WebMD Global LLC – Lecture fees.
Vipul Jairath, MBChB, DPhil, MRCP1, Britta Siegmund, MD2, Lindsey Surace, MD3, Ngozi Erondu, MD, PhD3, Minhu Chen, MD, PhD4, Karen Chachu, MD, PhD5, Edouard Louis, MD, PhD6, Katsuyoshi Matsuoka, MD, PhD7, Jimmy Limdi, MD8, Edmund Arthur, PharmD, MBA3, Nicole Houck, BS3, Mary Ellen Frustaci, BS, MS3, Joyce Zhan, PhD3, Grazyna Rydzewska, MD, PhD9, Edward V.. Loftus, MD10, Maria T.. Abreu, MD11, 65, Efficacy and Safety of Icotrokinra, a Targeted Oral Peptide That Selectively Blocks IL-23 Receptor Activation, in Ulcerative Colitis: Results From Week 28 of ANTHEM-UC, a Phase 2b Dose-Ranging Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1Western University, London, ON, Canada; 2Charité – Universitätsmedizin Berlin, corporate member of Freie Universität Berlin and Humboldt-Universität zu Berlin, Berlin, Berlin, Germany; 3Johnson & Johnson, Spring House, PA; 4The First Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, China; 5Duke University School of Medicine, Durham, NC; 6CHU Liège University Hospital, Liège, Liege, Belgium; 7Toho University Sakura Medical Center, Chiba, Chiba, Japan; 8Northern Care Alliance NHS Foundation Trust and University of Manchester, Manchester, England, United Kingdom; 9National Medical Institute of Ministry of Interior and Administration, Warsaw, Mazowieckie, Poland; 10Mayo Clinic College of Medicine and Science and Mayo Clinic, Rochester, MN; 11Cedars-Sinai, Los Angeles, CA
Introduction: As previously reported, all 3 doses of icotrokinra (ICO; JNJ-2113) — a first-in-class targeted oral peptide that selectively blocks IL-23 receptor activation — met the week (W) 12 primary endpoint in ANTHEM-UC, a Phase 2b, randomized, double-blind, placebo-controlled, treat-through, dose-ranging study in adults with moderate to severe ulcerative colitis (UC). Here we report W28 efficacy and safety of ICO from ANTHEM-UC.
Methods: Participants (pts) had a modified Mayo score (mMS) of 5–9 inclusive, a Mayo endoscopy subscore (MES) ≥2, and inadequate response/intolerance (IR) to TNFα blockers, vedolizumab, ustekinumab, JAK inhibitors, or S1P receptor modulators (BIO/JAKi/S1P-IR) or IR to corticosteroids, AZA, or 6-MP. Randomization (1:1:1:1) to once-daily (qd) oral ICO 100 mg, 200 mg, 400 mg or placebo (PBO) was stratified by BIO/JAKi/S1P-IR status (Y/N) and MES (2 or 3).
Clinical response at W12 was the primary endpoint. Clinical response, clinical remission, symptomatic remission, endoscopic improvement, and histologic-endoscopic mucosal improvement (HEMI) were further evaluated at W28 as exploratory endpoints. Pts meeting inadequate response criteria at W16 had a treatment adjustment: PBO pts switched to ICO 400 mg qd and ICO pts had a sham adjustment. All pts meeting inadequate response criteria were considered non-responders moving forward.
Results: Analyses included all 252 randomized pts who received study medication: mean mMS, 6.63; mMS >7, 31.9%; MES=3, 58.7%; BIO/JAKi/S1P-IR, 43.3%. W16 inadequate response criteria were met by 24 (38.1%), 11 (17.2%), 11 (17.7%) and 7 (11.1%) pts receiving PBO & ICO 100, 200 and 400 mg.
Relative to PBO, all ICO doses demonstrated clinically meaningful rates of clinical response, clinical remission, symptomatic remission, endoscopic improvement, and HEMI at W28 (Figure). Relative to W12 outcomes, rates of clinical response, clinical remission, endoscopic improvement, and HEMI continued to increase through W28 in each ICO dose group.
Through W28, ≥1 AE (61.9%, 65.6%, 66.1%, 60.3%), SAEs (9.5%, 0%, 4.8%, 1.6%), or AEs leading to discontinuation of study agent (11.1%, 0%, 6.5%, 3.2%) were reported in pts in the PBO & ICO 100, 200 and 400 mg groups. No serious or opportunistic infections, tuberculosis, malignancies, clinically important hepatic disorders, VTE, MACE, or deaths were reported with ICO.
Discussion: In ANTHEM-UC, clinical efficacy and a favorable safety profile were observed with once-daily ICO through W28 in pts with moderate to severe UC.

Figure: Week 28 Outcomes in ANTHEM-UC:
Data presented as percentage of participants attaining the endpoint, with adjusted treatment difference (Δ), 95% confidence interval (CI) for the adjusted treatment difference, and p-value versus placebo. At Week 28, all endpoints are exploratory and p-values are nominal. Outcomes at Week 12 that reached statistical significance per the prespecified testing procedure are indicated.
Adjusted treatment differences, 95% CIs, and p-values were based on the common risk difference by use of Mantel-Haenszel stratum weights and the Sato variance estimator, using stratification factors of inadequate response/intolerance to TNF blockers, vedolizumab, ustekinumab, JAK inhibitors, or S1P receptor modulators (Yes or No) and Mayo endoscopy subscore (MES) (2 [moderate] or 3 [severe]).
Participants with intercurrent events of ostomy or colectomy, prohibited changes in UC medication, discontinuation of study intervention for any reason except those due to major disruptions (e.g., COVID-19 related reasons or regional crisis, excluding COVID-19 infection), or meeting Week 16 inadequate response criteria were considered nonresponders for the Week 28 endpoints, as were participants who had missing data for the endpoint.
ENDPOINT DEFINITIONS: a. Clinical response: a decrease from baseline in modified Mayo score by ≥30% and ≥2 points with either a ≥1-point decrease from baseline in rectal bleeding subscore or a rectal bleeding subscore of 0 or 1. b. Clinical remission: stool frequency subscore of 0 or 1, rectal bleeding subscore of 0, and MES of 0 or 1. c. Symptomatic remission: stool frequency subscore of 0 or 1 and rectal bleeding subscore of 0
d. Endoscopic improvement: MES of 0 or 1. e. Histologic-endoscopic mucosal improvement (HEMI): histologic remission (absence of neutrophils from the mucosa [both lamina propria and epithelium], no crypt destruction, and no erosions, ulcerations, or granulation tissue according to the Geboes grading system) AND endoscopic improvement (MES of 0 or 1).
Disclosures:
Vipul Jairath: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Alimentiv – Consultant, Grant/Research Support. Arena Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Asahi Kasei Pharma – Consultant, Grant/Research Support, Speakers Bureau. Asieris Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. AstraZeneca – Consultant, Grant/Research Support, Speakers Bureau. Avoro Capital – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, Speakers Bureau. Celltrion – Consultant, Grant/Research Support, Speakers Bureau. Eli Lilly and Company – Consultant, Grant/Research Support, Speakers Bureau. Endpoint Health – Consultant. Enthera – Consultant. Ferring Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Flagship Pioneering – Consultant, Grant/Research Support, Speakers Bureau. Fresenius Kabi – Consultant, Grant/Research Support, Speakers Bureau. Galapagos NV – Consultant, Grant/Research Support, Speakers Bureau. Genentech – Consultant, Grant/Research Support, Speakers Bureau. Gilde Healthcare – Consultant. Gilead Sciences – Consultant, Grant/Research Support, Speakers Bureau. GlaxoSmithKline – Consultant, Grant/Research Support, Speakers Bureau. Innomar – Consultant. JAMP – Consultant. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Merck – Consultant, Grant/Research Support, Speakers Bureau. Metacrine – Consultant, Grant/Research Support, Speakers Bureau. Mylan – Consultant, Grant/Research Support, Speakers Bureau. Pandion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Pendopharm – Consultant, Grant/Research Support, Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. Prometheus Therapeutics and Diagnostics – Consultant, Grant/Research Support, Speakers Bureau. Protagonist Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Reistone Biopharma – Consultant, Grant/Research Support, Speakers Bureau. Roche – Consultant, Grant/Research Support, Speakers Bureau. Roivant – Consultant. Sandoz – Consultant, Grant/Research Support, Speakers Bureau. Second Genome – Consultant, Grant/Research Support, Speakers Bureau. Shire – Speakers Bureau. Sorriso Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Syndegen – Consultant. Takeda – Consultant, Intellectual Property/Patents, Speakers Bureau. TD Securities – Consultant. Teva – Consultant, Grant/Research Support, Speakers Bureau. Topivert – Consultant, Grant/Research Support, Speakers Bureau. Ventyx Biosciences – Consultant, Grant/Research Support, Speakers Bureau. Vividion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau.
Britta Siegmund: AbbVie – Consultant, Speakers Bureau. Abivax – Consultant. AlfaSigma – Speakers Bureau. Boehringer Ingelheim – Consultant. Bristol Myers Squibb – Consultant, Speakers Bureau. CED Service GmbH – Speakers Bureau. Dr. Falk Pharma – Consultant, Speakers Bureau. Eli Lilly – Consultant, Speakers Bureau. Endpoint Health – Consultant. Ferring – Speakers Bureau. Galapagos – Consultant, Speakers Bureau. Gilead – Consultant. Johnson & Johnson – Consultant, Speakers Bureau. Landos – Consultant. Materia Prima – Consultant. MSD – Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. PredictImmune – Consultant. Takeda – Consultant, Speakers Bureau. Tr1X – Speakers Bureau.
Lindsey Surace: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Ngozi Erondu: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Minhu Chen: AbbVie – Provided Educational Activities. Boehringer Ingelheim – Advisory Committee/Board Member. China Medical System – Provided Educational Activities. IPSEN – Provided Educational Activities. Johnson & Johnson – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Takeda – Grant/Research Support, Provided Educational Activities.
Karen Chachu: Johnson & Johnson – Advisor or Review Panel Member. OptumRx – Consultant.
Edouard Louis: AbbVie – Advisor or Review Panel Member, Consultant, Grant/Research Support, Speakers Bureau. Aboleris – Consultant. Biokuris – Consultant. BMS – Advisor or Review Panel Member, Speakers Bureau. Celgene – Advisory Committee/Board Member. Celltrion – Grant/Research Support, Speakers Bureau. Eli Lilly – Advisory Committee/Board Member. Falk – Grant/Research Support. Ferring – Advisor or Review Panel Member, Speakers Bureau. Fresenius-Kabi – Grant/Research Support, Speakers Bureau. Galapagos – Speakers Bureau. Johnson & Johnson – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Pfizer – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Takeda – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Thabor – Consultant.
Katsuyoshi Matsuoka: AbbVie – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Bristol Myers Squibb – Advisor or Review Panel Member. Celltrion Healthcare – Advisor or Review Panel Member. EA Pharma – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Eli Lilly – Advisor or Review Panel Member, Speakers Bureau. Gilead – Advisor or Review Panel Member, Speakers Bureau. JIMRO – Grant/Research Support. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Kyorin – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Mitsubishi Tanabe Pharma – Grant/Research Support, Speakers Bureau. Mochida – Advisor or Review Panel Member, Grant/Research Support, Speakers Bureau. Nippon Kayaku – Grant/Research Support, Speakers Bureau. Takeda – Advisor or Review Panel Member, Speakers Bureau. Zeria – Grant/Research Support, Speakers Bureau.
Jimmy Limdi: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Abivax – Consultant, Speakers Bureau. AlfaSigma – Consultant, Speakers Bureau. Biohit – Consultant, Speakers Bureau. Bristol Myers Squibb – Consultant, Speakers Bureau. Celltrion – Consultant, Speakers Bureau. Eli Lilly and Company – Consultant, Speakers Bureau. Ferring – Consultant, Speakers Bureau. Galapagos – Consultant, Grant/Research Support, Speakers Bureau. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. MSD – Consultant, Speakers Bureau. Pfizer – Consultant, Speakers Bureau. Takeda – Consultant, Grant/Research Support, Speakers Bureau.
Edmund Arthur: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Nicole Houck: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Mary Ellen Frustaci: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Joyce Zhan: Johnson & Johnson – Employee, Stock-publicly held company(excluding mutual/index funds).
Grazyna Rydzewska: AbbVie – Consultant, travel/accommodation meeting expenses, Speakers Bureau. AlfaSigma – Consultant, Speakers Bureau. AstraZeneca – Consultant, Speakers Bureau. Bayer – Consultant, Speakers Bureau. Biocodex – Consultant, Speakers Bureau. Bristol Myers Squibb – Consultant, Speakers Bureau. Ferring – Consultant, travel/accommodation meeting expenses, Speakers Bureau. Johnson & Johnson – Consultant, Speakers Bureau. Lilly – Consultant, Speakers Bureau. PRO.MED.Pl – Consultant, Speakers Bureau. Recordati – Consultant, Speakers Bureau. Sanprobi – Consultant, Speakers Bureau. SOBI – Consultant, Speakers Bureau. Takeda – Consultant, travel/accommodation meeting expenses, Speakers Bureau.
Edward Loftus: AbbVie – Consultant, Grant/Research Support. Abivax – Consultant. Astellas – Consultant. Avalo – Consultant. Biocon – Consultant. Celltrion – Consultant. Eli Lilly – Advisory Committee/Board Member, Consultant. Exact Sciences – Stock Options. Fresenius Kabi – Consultant. Genentech – Advisory Committee/Board Member. Gilead – Consultant, Grant/Research Support. Iota Biosciences – Consultant. Iterative Health – Consultant. Johnson & Johnson – Consultant, Grant/Research Support. Merck – Consultant, Grant/Research Support. Moderna – Stock Options. Morphic – Consultant. Ono Pharma – Consultant. Spyre – Advisory Committee/Board Member. Takeda – Consultant, Grant/Research Support. TR1X Bio – Consultant.
Maria Abreu: AbbVie – Consultant. Alimentiv – Lecture fees. Arena Pharmaceuticals – Consultant. Bristol Myers Squibb – Consultant. Celsius Therapeutics – Consultant. Gilead – Consultant. Johnson & Johnson – Advisory Committee/Board Member, Lecture fees. Lilly – Consultant. Pfizer – Consultant. Prime – Lecture fees. Prometheus Biosciences – Consultant. UCB – Consultant. WebMD Global LLC – Lecture fees.
Vipul Jairath, MBChB, DPhil, MRCP1, Britta Siegmund, MD2, Lindsey Surace, MD3, Ngozi Erondu, MD, PhD3, Minhu Chen, MD, PhD4, Karen Chachu, MD, PhD5, Edouard Louis, MD, PhD6, Katsuyoshi Matsuoka, MD, PhD7, Jimmy Limdi, MD8, Edmund Arthur, PharmD, MBA3, Nicole Houck, BS3, Mary Ellen Frustaci, BS, MS3, Joyce Zhan, PhD3, Grazyna Rydzewska, MD, PhD9, Edward V.. Loftus, MD10, Maria T.. Abreu, MD11, 65, Efficacy and Safety of Icotrokinra, a Targeted Oral Peptide That Selectively Blocks IL-23 Receptor Activation, in Ulcerative Colitis: Results From Week 28 of ANTHEM-UC, a Phase 2b Dose-Ranging Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.

