Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 2A: Colon / IBD / Practice Management
26 - Both Low (TECH) and High (TEAM) Touch Interventions Improve Behavioral Health, IBD Symptoms, and Quality of Life: A Randomized Control Trial
Tuesday, October 28, 2025
9:40 AM - 9:50 AM PDT
Location: North Ballroom 120D

Laurie Keefer, PhD, FACG (she/her/hers)
Icahn School of Medicine at Mount Sinai
New York, NY
Presenting Author(s)
Laurie Keefer, PhD, FACG1, Benjamin L. Cohen, MD2, Jeffrey M. Dueker, MD, MPH3, Benjamin Click, MD, MS4, Marla C. Dubinsky, MD5, Miguel Regueiro, MD6, Stephen E. Lupe, PhD7, Ashley L. Taylor, MLIS8, Jane Kogan, PhD9, Cara Nikolajski, PhD, MPH3, Nina Oryshkewych, MS3, Meredith Wallace, PhD3, Eva Szigethy, MD, PhD, FACG10
1Icahn School of Medicine at Mount Sinai, New York, NY; 2Cleveland Clinic Foundation, Cleveland, OH; 3University of Pittsburgh, Pittsburgh, PA; 4University of Colorado, Denver, CO; 5Susan and Leonard Feinstein IBD Center, Icahn School of Medicine at Mount Sinai, New York, NY, USA, New York, NY; 6Cleveland Clinic, Cleveland, OH; 7The Cleveland Clinic, Hudson, OH; 8UPMC Center for High-Valure Health Care, Pittsburgh, PA; 9UPMC Center for High-Value Health Care, PIttsburgh, PA; 10Akron Childrens Hospital, Akron, OH
Introduction: Patients with inflammatory bowel disease (IBD) can have impaired mental and physical health, decreased quality of life, and increased healthcare services utilization related to their IBD. The specialty medical home (SMH) model, which integrates physical, nutritional, and psychosocial treatment for a patient-centered approach, has the potential to provide whole-person care that improves IBD and behavioral health (BH) symptom severity and other patient-centered outcomes.
Methods: Patients aged 18-60 with an IBD diagnosis and moderate-to-severe anxiety and depression across three academic centers were randomized to one of two evidence-based SMH care approaches. TECH participants received a low-human touch intervention supported by use of a digital cognitive behavioral therapy (dCBT) mobile application (app) with embedded health coaches, while TEAM participants received high-human touch multidisciplinary treatment. Primary outcomes were intervention impact on BH (PHQ8 + GAD7 score) and IBD (complexity score) symptom severity, with secondary outcomes of quality of life, functional impairment, and disease self-efficacy, at 6 and 12 months. IBD complexity score was calculated by adding the value of the patient’s PRO-2 UC or CD to the cumulative raw PROMIS-GI score, with lower scores indicating lower symptom severity.
Results: 657 participants (330 TEAM and 327 TECH) enrolled in the study between July 2019 and March 2025. Participants were mostly female (65%) and diagnosed with Crohn’s disease (67%) with no significant differences between the groups. Mean age (34.7, SD 10.7) and disease duration (10.4 years, SD 8.3) were similar between groups. Participants in each study group had significant improvements (p< 0.001) in both BH (Figure 1a) and IBD (Figure 1b) symptom severity after 6 months in the study, which was sustained at 12 months. Additionally, participants saw improvements in quality of life, functional impairment, and self-efficacy (Table 1).
Discussion: In the first large, randomized effectiveness trial comparing low (TECH) vs high (TEAM) human touch behavioral interventions within a SMH, patients in both groups demonstrated significant improvement in BH and IBD symptom severity as well as quality of life, functional, and self-efficacy outcomes. Further work is ongoing to identify which patients may benefit most from which interventions.
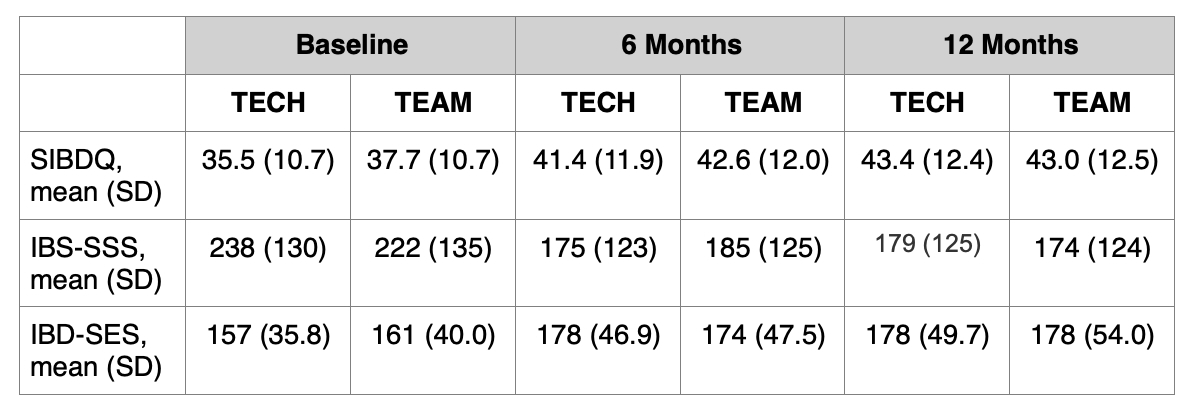
Figure: Figure 1a and 1b: Behavioral Health and IBD Symptom Severity Scores by Intervention Group Over Time
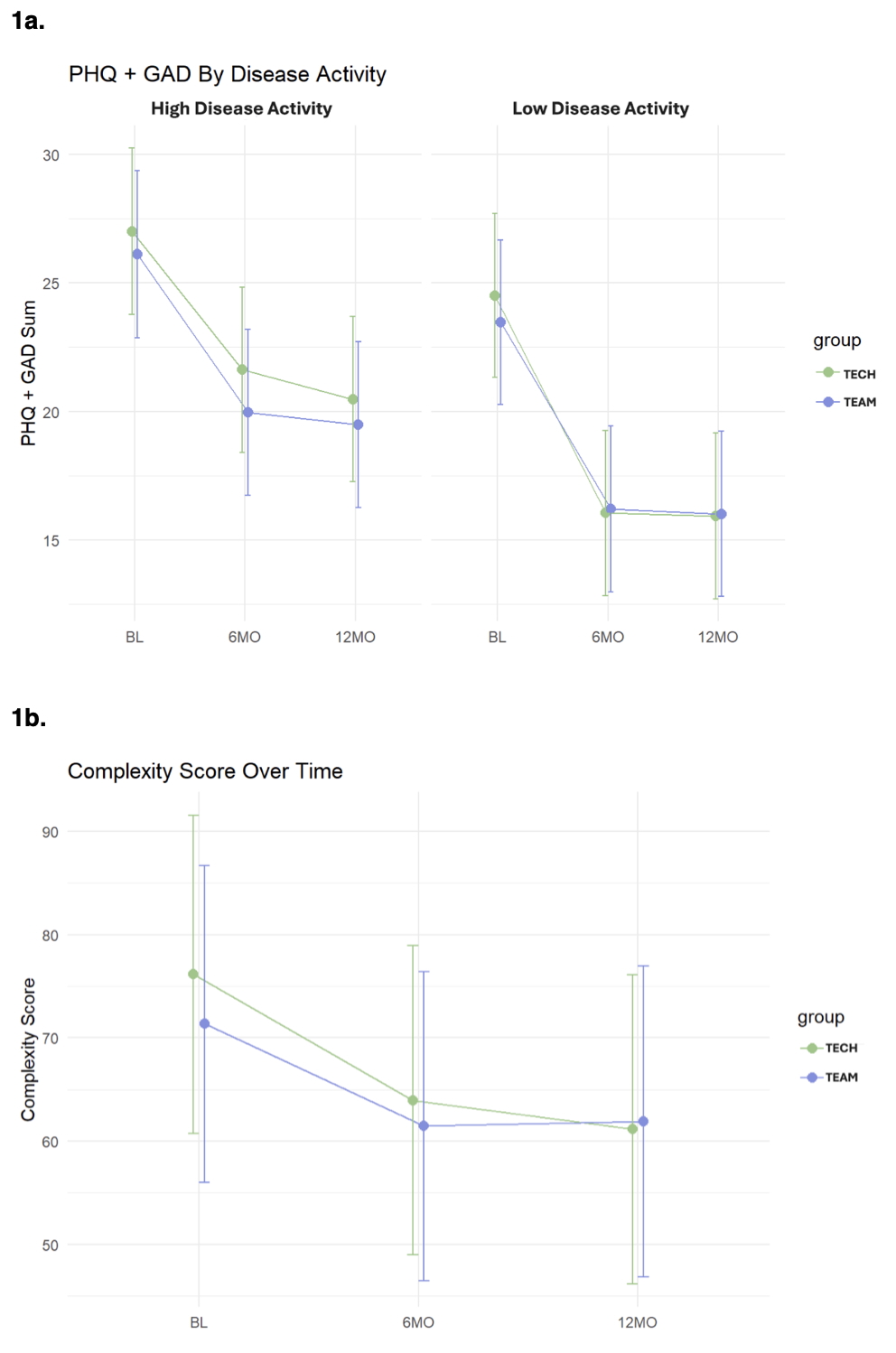
Figure: Table 1: Quality of life, functional, and self-efficacy outcomes at 6 and 12 months. Abbreviations: Short Quality of Life in IBD Questionnaire (SIBDQ); Irritable Bowel Syndrome Symptom Severity Score (IBS-SSS); IBD Self-Efficacy Scale (IBD-SES)
Disclosures:
Laurie Keefer: AbbVie – Consultant. Ardelyx – Consultant. Eli Lilly – Consultant. Janssen – Consultant. Pfizer – Consultant. Reckitt Health – Consultant. Trellus Health – Owner/Ownership Interest, Stock-publicly held company(excluding mutual/index funds).
Benjamin Cohen: Abbvie – Advisory Committee/Board Member, Consultant, Speakers Bureau. ALPCO – Advisory Committee/Board Member, Consultant. Emmes Biopharma Services LLC – DSMB. J&J Innovative Medicine – Advisory Committee/Board Member. Pfizer – Advisory Committee/Board Member. Takeda – Consultant, Speakers Bureau.
Jeffrey Dueker indicated no relevant financial relationships.
Benjamin Click indicated no relevant financial relationships.
Marla Dubinsky: AbbVie – Consultant, Grant/Research Support. Arena – Consultant. Astra Zeneca – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Eli Lilly – Consultant. Gilead – Consultant. Janssen – Consultant, Grant/Research Support. Merck – Consultant. Pfizer – Consultant. Prometheus Laboratories – Consultant, Grant/Research Support. Sanofi – Consultant. Spyre – Consultant. Takeda – Consultant. Trellus Health – Stock Options, Stock-publicly held company(excluding mutual/index funds). UCB – Consultant.
Miguel Regueiro: AbbVie – Advisory Committee/Board Member, Consultant. Amgen – Advisory Committee/Board Member, Consultant. BMS – Advisory Committee/Board Member, Consultant. Boehringer Ingelheim Pharmaceuticals Inc. – Advisory Committee/Board Member, Consultant. Celegene – Advisory Committee/Board Member, Consultant. Eli Lilly and Company – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Gilead – Advisory Committee/Board Member, Consultant. Janssen – Advisory Committee/Board Member, Consultant. Organon – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Prometheus – Advisory Committee/Board Member, Consultant. Salix – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB – Advisory Committee/Board Member, Consultant.
Stephen Lupe: Ayble Health – Advisory Committee/Board Member. Boomerang Health – Advisory Committee/Board Member. Takeda – Consultant, Speakers Bureau.
Ashley Taylor indicated no relevant financial relationships.
Jane Kogan indicated no relevant financial relationships.
Cara Nikolajski indicated no relevant financial relationships.
Nina Oryshkewych indicated no relevant financial relationships.
Meredith Wallace indicated no relevant financial relationships.
Eva Szigethy: PCORI – Grant/Research Support.
Laurie Keefer, PhD, FACG1, Benjamin L. Cohen, MD2, Jeffrey M. Dueker, MD, MPH3, Benjamin Click, MD, MS4, Marla C. Dubinsky, MD5, Miguel Regueiro, MD6, Stephen E. Lupe, PhD7, Ashley L. Taylor, MLIS8, Jane Kogan, PhD9, Cara Nikolajski, PhD, MPH3, Nina Oryshkewych, MS3, Meredith Wallace, PhD3, Eva Szigethy, MD, PhD, FACG10, 26, Both Low (TECH) and High (TEAM) Touch Interventions Improve Behavioral Health, IBD Symptoms, and Quality of Life: A Randomized Control Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1Icahn School of Medicine at Mount Sinai, New York, NY; 2Cleveland Clinic Foundation, Cleveland, OH; 3University of Pittsburgh, Pittsburgh, PA; 4University of Colorado, Denver, CO; 5Susan and Leonard Feinstein IBD Center, Icahn School of Medicine at Mount Sinai, New York, NY, USA, New York, NY; 6Cleveland Clinic, Cleveland, OH; 7The Cleveland Clinic, Hudson, OH; 8UPMC Center for High-Valure Health Care, Pittsburgh, PA; 9UPMC Center for High-Value Health Care, PIttsburgh, PA; 10Akron Childrens Hospital, Akron, OH
Introduction: Patients with inflammatory bowel disease (IBD) can have impaired mental and physical health, decreased quality of life, and increased healthcare services utilization related to their IBD. The specialty medical home (SMH) model, which integrates physical, nutritional, and psychosocial treatment for a patient-centered approach, has the potential to provide whole-person care that improves IBD and behavioral health (BH) symptom severity and other patient-centered outcomes.
Methods: Patients aged 18-60 with an IBD diagnosis and moderate-to-severe anxiety and depression across three academic centers were randomized to one of two evidence-based SMH care approaches. TECH participants received a low-human touch intervention supported by use of a digital cognitive behavioral therapy (dCBT) mobile application (app) with embedded health coaches, while TEAM participants received high-human touch multidisciplinary treatment. Primary outcomes were intervention impact on BH (PHQ8 + GAD7 score) and IBD (complexity score) symptom severity, with secondary outcomes of quality of life, functional impairment, and disease self-efficacy, at 6 and 12 months. IBD complexity score was calculated by adding the value of the patient’s PRO-2 UC or CD to the cumulative raw PROMIS-GI score, with lower scores indicating lower symptom severity.
Results: 657 participants (330 TEAM and 327 TECH) enrolled in the study between July 2019 and March 2025. Participants were mostly female (65%) and diagnosed with Crohn’s disease (67%) with no significant differences between the groups. Mean age (34.7, SD 10.7) and disease duration (10.4 years, SD 8.3) were similar between groups. Participants in each study group had significant improvements (p< 0.001) in both BH (Figure 1a) and IBD (Figure 1b) symptom severity after 6 months in the study, which was sustained at 12 months. Additionally, participants saw improvements in quality of life, functional impairment, and self-efficacy (Table 1).
Discussion: In the first large, randomized effectiveness trial comparing low (TECH) vs high (TEAM) human touch behavioral interventions within a SMH, patients in both groups demonstrated significant improvement in BH and IBD symptom severity as well as quality of life, functional, and self-efficacy outcomes. Further work is ongoing to identify which patients may benefit most from which interventions.

Figure: Figure 1a and 1b: Behavioral Health and IBD Symptom Severity Scores by Intervention Group Over Time

Figure: Table 1: Quality of life, functional, and self-efficacy outcomes at 6 and 12 months. Abbreviations: Short Quality of Life in IBD Questionnaire (SIBDQ); Irritable Bowel Syndrome Symptom Severity Score (IBS-SSS); IBD Self-Efficacy Scale (IBD-SES)
Disclosures:
Laurie Keefer: AbbVie – Consultant. Ardelyx – Consultant. Eli Lilly – Consultant. Janssen – Consultant. Pfizer – Consultant. Reckitt Health – Consultant. Trellus Health – Owner/Ownership Interest, Stock-publicly held company(excluding mutual/index funds).
Benjamin Cohen: Abbvie – Advisory Committee/Board Member, Consultant, Speakers Bureau. ALPCO – Advisory Committee/Board Member, Consultant. Emmes Biopharma Services LLC – DSMB. J&J Innovative Medicine – Advisory Committee/Board Member. Pfizer – Advisory Committee/Board Member. Takeda – Consultant, Speakers Bureau.
Jeffrey Dueker indicated no relevant financial relationships.
Benjamin Click indicated no relevant financial relationships.
Marla Dubinsky: AbbVie – Consultant, Grant/Research Support. Arena – Consultant. Astra Zeneca – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Eli Lilly – Consultant. Gilead – Consultant. Janssen – Consultant, Grant/Research Support. Merck – Consultant. Pfizer – Consultant. Prometheus Laboratories – Consultant, Grant/Research Support. Sanofi – Consultant. Spyre – Consultant. Takeda – Consultant. Trellus Health – Stock Options, Stock-publicly held company(excluding mutual/index funds). UCB – Consultant.
Miguel Regueiro: AbbVie – Advisory Committee/Board Member, Consultant. Amgen – Advisory Committee/Board Member, Consultant. BMS – Advisory Committee/Board Member, Consultant. Boehringer Ingelheim Pharmaceuticals Inc. – Advisory Committee/Board Member, Consultant. Celegene – Advisory Committee/Board Member, Consultant. Eli Lilly and Company – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Gilead – Advisory Committee/Board Member, Consultant. Janssen – Advisory Committee/Board Member, Consultant. Organon – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Prometheus – Advisory Committee/Board Member, Consultant. Salix – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB – Advisory Committee/Board Member, Consultant.
Stephen Lupe: Ayble Health – Advisory Committee/Board Member. Boomerang Health – Advisory Committee/Board Member. Takeda – Consultant, Speakers Bureau.
Ashley Taylor indicated no relevant financial relationships.
Jane Kogan indicated no relevant financial relationships.
Cara Nikolajski indicated no relevant financial relationships.
Nina Oryshkewych indicated no relevant financial relationships.
Meredith Wallace indicated no relevant financial relationships.
Eva Szigethy: PCORI – Grant/Research Support.
Laurie Keefer, PhD, FACG1, Benjamin L. Cohen, MD2, Jeffrey M. Dueker, MD, MPH3, Benjamin Click, MD, MS4, Marla C. Dubinsky, MD5, Miguel Regueiro, MD6, Stephen E. Lupe, PhD7, Ashley L. Taylor, MLIS8, Jane Kogan, PhD9, Cara Nikolajski, PhD, MPH3, Nina Oryshkewych, MS3, Meredith Wallace, PhD3, Eva Szigethy, MD, PhD, FACG10, 26, Both Low (TECH) and High (TEAM) Touch Interventions Improve Behavioral Health, IBD Symptoms, and Quality of Life: A Randomized Control Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.


