Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
71 - Real-World Comparative Effectiveness of Vedolizumab versus Upadacitinib and Risankizumab for Crohn’s Disease Through 52 Weeks
Wednesday, October 29, 2025
9:30 AM - 9:40 AM PDT
Location: North Ballroom 120BC

Rahul S. Dalal, MD, MPH (he/him/his)
Brigham and Women's Hospital, Harvard Medical School
Boston, MA
Presenting Author(s)
Rahul S. Dalal, MD, MPH1, Grace Hardwick, BS1, Lindsay Clarke, MD1, Heidy Cabral, MS, BS1, Alexander Carlin, BS1, Jessica R.. Allegretti, MD, MPH2
1Brigham and Women's Hospital, Harvard Medical School, Boston, MA; 2Brigham and Women’s Hospital, Harvard Medical School, Boston, MA
Introduction: In a previous cohort study, upadacitinib (UPA) and risankizumab (RISA) demonstrated similar effectiveness for Crohn’s disease (CD). We now compare vedolizumab (VEDO) to UPA and RISA.
Methods: In this retrospective cohort study, adults initiated VEDO, UPA, or RISA for active CD at a large academic center in 2023. Pts were excluded if treatment was to prevent post-op recurrence or if the Harvey-Bradshaw Index was < 5 (unless on corticosteroids). Co-primary outcomes were steroid-free clinical remission (SFCR) and treatment discontinuation at 52 (+/-4) wks. Secondary outcomes were SFCR at 12 (+/-4) wks, clinical response (CR) at 12 and 52 (+/-4) wks, and endoscopic/radiologic response. Additional descriptive outcomes were bowel resection surgeries and adverse events (AEs). Outcome definitions are in the Fig 1 caption. Treatment discontinuation was evaluated using Cox regression and competing-risks regression, where discontinuation due to AEs was treated as a competing risk. Other outcomes were evaluated using logistic regression. Inverse probability of treatment weighting (IPTW) adjusted for confounders. Two independent comparisons were conducted: VEDO vs UPA and VEDO vs RISA.
Results: Among 291 pts, 72 received VEDO, 67 UPA, and 152 RISA. VEDO pts were younger, had less penetrating disease, and had fewer prior biologics (Table 1A). Unadjusted outcomes were similar for VEDO vs UPA (Fig 1A), but RISA had higher CR at 52 wks (80% vs 60%, p< 0.01), fewer discontinuations (18% vs 32%, p=0.02), and fewer AEs (20% vs 33%, p=0.04) vs VEDO. IPTW confirmed covariate balance (Table 1B). Competing risks regression showed similar discontinuation for non-response for VEDO vs UPA (Fig 1C), but higher discontinuation for VEDO vs RISA (Fig 1D). In IPTW models (Fig 1D&E), VEDO had lower odds of CR at 12 wks vs UPA (OR 0.36, p=0.02). Compared to RISA, VEDO had lower odds of CR at 12 wks (OR 0.51, p=0.04), CR at 52 wks (OR 0.32, p< 0.01), SFCR at 52 wks (OR 0.44, p=0.01), and endoscopic response (OR 0.28, p=0.01), and higher hazard of treatment discontinuation (HR 2.18, p=0.01). Common AEs: VEDO, infections (12/24), arthralgias (6/24); UPA, rashes (10/26), infections (7/26); RISA, infections (13/32), rashes (5/32).
Discussion: UPA was more likely than VEDO to achieve CR at 12 wks, but no differences were observed at 52 wks, possibly due to power. RISA was more effective than VEDO at both 12 and 52 wks. No new safety concerns emerged. Large, prospective studies are needed to confirm these findings.
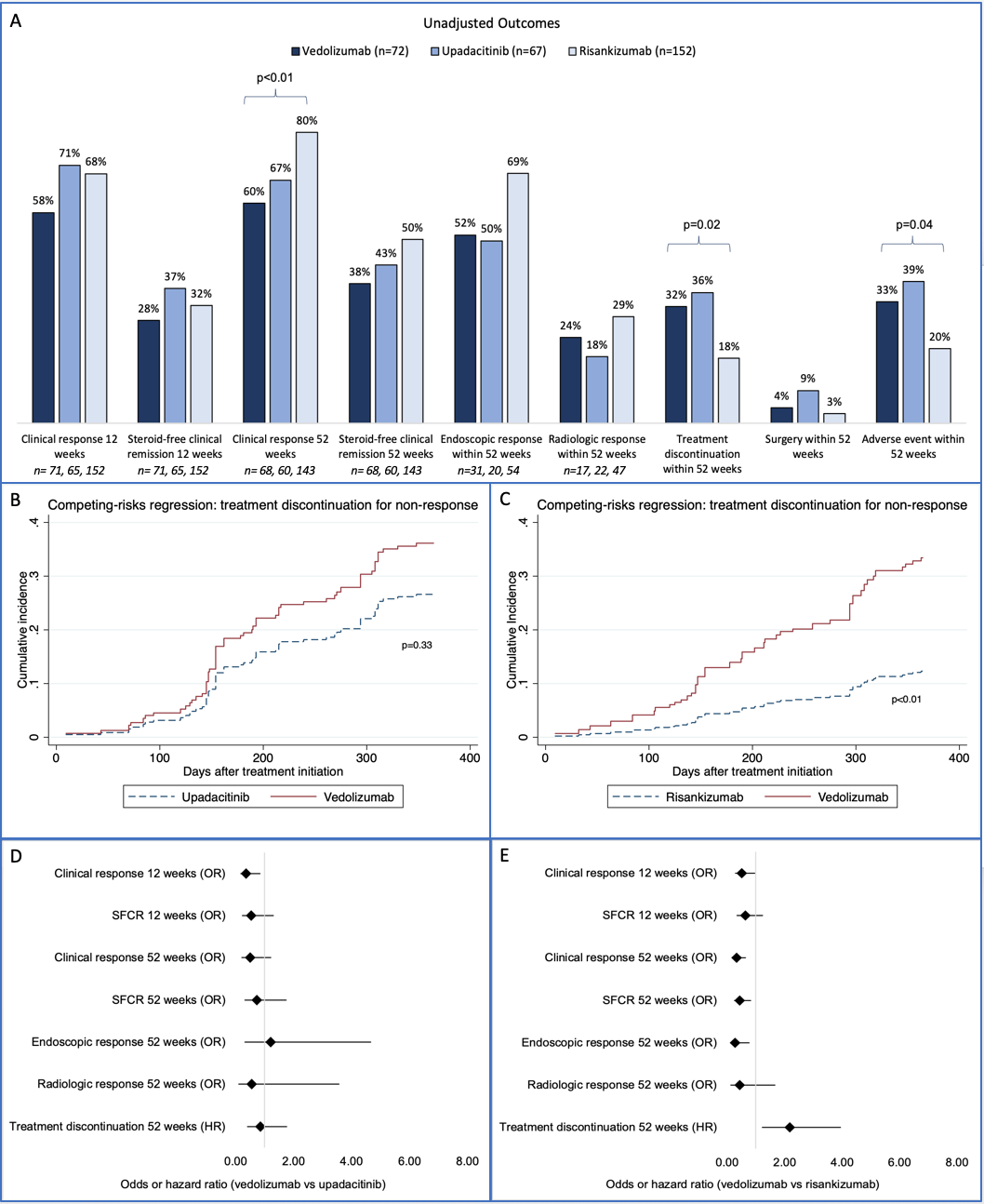
Figure: *Sample sizes are specified for variables where data are not available for all patients in cohort.
Abbreviations: IQR=interquartile range, BMI=body mass index, CD=Crohn’s disease, VEDO=vedolizumab, UPA=upadacitinib, RISA=risankizumab, IPTW=inverse probability of treatment weighting
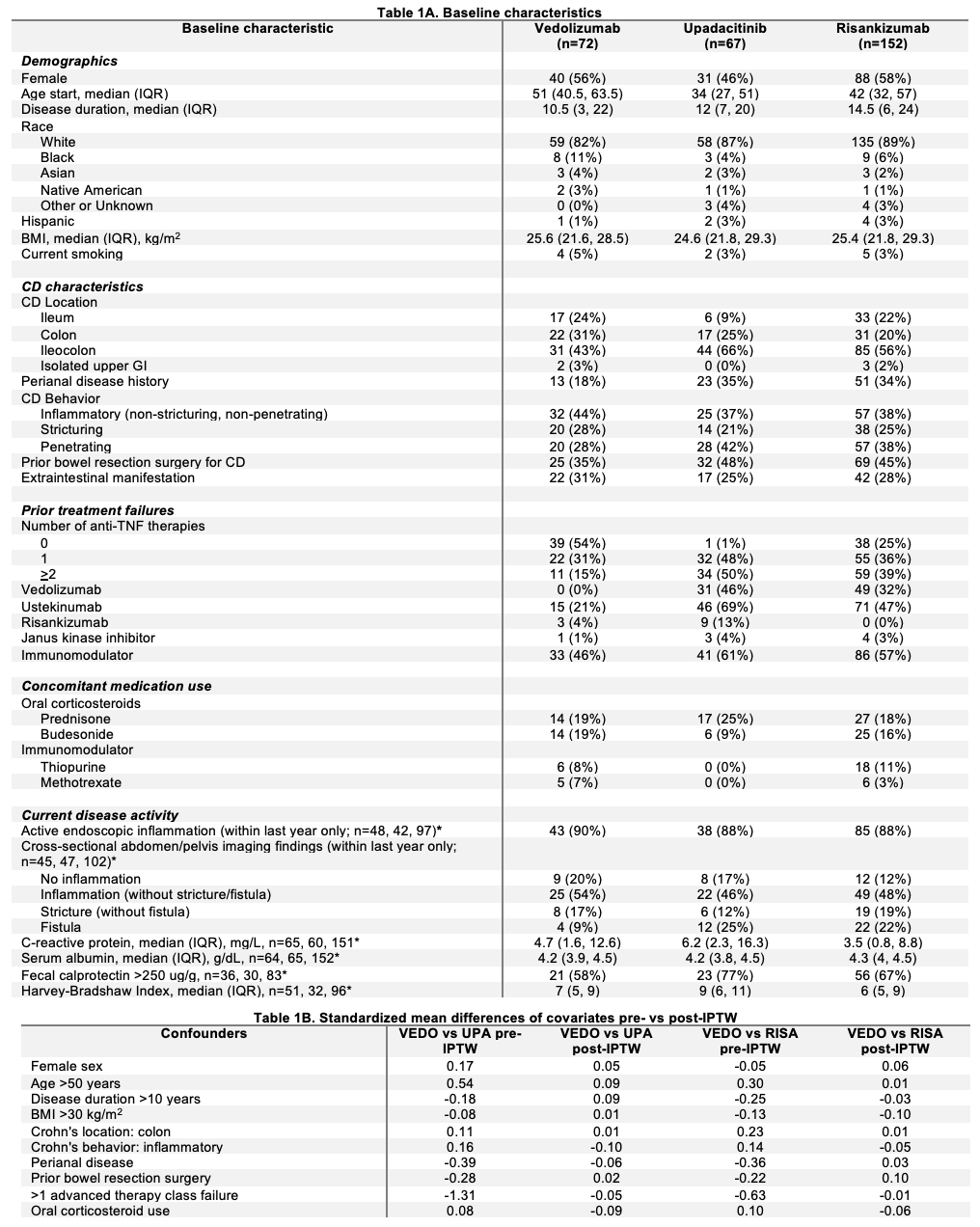
Figure: Figure 1. A. Unadjusted outcomes. In cases of missing data, sample sizes are indicated at bottom of graph. Clinical response = reduction in Harvey-Bradshaw Index (HBI) by >3 pts. Steroid-free clinical remission = HBI <5 with no use of corticosteroids at follow-up. Endoscopic response = simple endoscopic score for CD (SES-CD) reduction of 50%. Radiologic response = improved CD findings without new findings of active CD per radiologist’s assessment of cross-sectional imaging. Reference endoscopy/imaging studies were required to be within 1 year prior to treatment initiation. Provider and endoscopist global assessments were utilized when HBI and SES-CD were not documented or applicable. B and C. Competing risk regression analyses with weighted cumulative incidence curves for treatment discontinuation due to non-response for vedolizumab vs upadacitinib (B) and vedolizumab vs risankizumab (C). Curves are adjusted for treatment discontinuation due to adverse events as a competing event. D and E. IPTW logistic and Cox regression analysis results for vedolizumab vs upadacitinib (D) and vedolizumab vs risankizumab (E).
Abbreviations: OR=odds ratio, HR=hazard ratio
Disclosures:
Rahul Dalal: Janssen – Grant/Research Support. Takeda – Consultant.
Grace Hardwick indicated no relevant financial relationships.
Lindsay Clarke indicated no relevant financial relationships.
Heidy Cabral indicated no relevant financial relationships.
Alexander Carlin indicated no relevant financial relationships.
Jessica Allegretti: Abbvie – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speaker, Speakers Bureau. Adiso – Consultant. Bristol Myer Squibb – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speaker. Celltrion – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Ferring – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Finch – Consultant. Genentech – Advisory Committee/Board Member, Consultant. GlaxoSmithKline – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Iterative Scopes – Consultant. Janssen – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speakers Bureau. Johnson & Johnson – Consultant, Grant/Research Support, Speaker. Merck – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Grant/Research Support. Pfizer – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Grant/Research Support. Roivant – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Roivant Adiso – Consultant. Seres Therapeutics – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Shattuck Labs – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. TRXBio – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Vedanta – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant.
Rahul S. Dalal, MD, MPH1, Grace Hardwick, BS1, Lindsay Clarke, MD1, Heidy Cabral, MS, BS1, Alexander Carlin, BS1, Jessica R.. Allegretti, MD, MPH2, 71, Real-World Comparative Effectiveness of Vedolizumab versus Upadacitinib and Risankizumab for Crohn’s Disease Through 52 Weeks, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1Brigham and Women's Hospital, Harvard Medical School, Boston, MA; 2Brigham and Women’s Hospital, Harvard Medical School, Boston, MA
Introduction: In a previous cohort study, upadacitinib (UPA) and risankizumab (RISA) demonstrated similar effectiveness for Crohn’s disease (CD). We now compare vedolizumab (VEDO) to UPA and RISA.
Methods: In this retrospective cohort study, adults initiated VEDO, UPA, or RISA for active CD at a large academic center in 2023. Pts were excluded if treatment was to prevent post-op recurrence or if the Harvey-Bradshaw Index was < 5 (unless on corticosteroids). Co-primary outcomes were steroid-free clinical remission (SFCR) and treatment discontinuation at 52 (+/-4) wks. Secondary outcomes were SFCR at 12 (+/-4) wks, clinical response (CR) at 12 and 52 (+/-4) wks, and endoscopic/radiologic response. Additional descriptive outcomes were bowel resection surgeries and adverse events (AEs). Outcome definitions are in the Fig 1 caption. Treatment discontinuation was evaluated using Cox regression and competing-risks regression, where discontinuation due to AEs was treated as a competing risk. Other outcomes were evaluated using logistic regression. Inverse probability of treatment weighting (IPTW) adjusted for confounders. Two independent comparisons were conducted: VEDO vs UPA and VEDO vs RISA.
Results: Among 291 pts, 72 received VEDO, 67 UPA, and 152 RISA. VEDO pts were younger, had less penetrating disease, and had fewer prior biologics (Table 1A). Unadjusted outcomes were similar for VEDO vs UPA (Fig 1A), but RISA had higher CR at 52 wks (80% vs 60%, p< 0.01), fewer discontinuations (18% vs 32%, p=0.02), and fewer AEs (20% vs 33%, p=0.04) vs VEDO. IPTW confirmed covariate balance (Table 1B). Competing risks regression showed similar discontinuation for non-response for VEDO vs UPA (Fig 1C), but higher discontinuation for VEDO vs RISA (Fig 1D). In IPTW models (Fig 1D&E), VEDO had lower odds of CR at 12 wks vs UPA (OR 0.36, p=0.02). Compared to RISA, VEDO had lower odds of CR at 12 wks (OR 0.51, p=0.04), CR at 52 wks (OR 0.32, p< 0.01), SFCR at 52 wks (OR 0.44, p=0.01), and endoscopic response (OR 0.28, p=0.01), and higher hazard of treatment discontinuation (HR 2.18, p=0.01). Common AEs: VEDO, infections (12/24), arthralgias (6/24); UPA, rashes (10/26), infections (7/26); RISA, infections (13/32), rashes (5/32).
Discussion: UPA was more likely than VEDO to achieve CR at 12 wks, but no differences were observed at 52 wks, possibly due to power. RISA was more effective than VEDO at both 12 and 52 wks. No new safety concerns emerged. Large, prospective studies are needed to confirm these findings.

Figure: *Sample sizes are specified for variables where data are not available for all patients in cohort.
Abbreviations: IQR=interquartile range, BMI=body mass index, CD=Crohn’s disease, VEDO=vedolizumab, UPA=upadacitinib, RISA=risankizumab, IPTW=inverse probability of treatment weighting

Figure: Figure 1. A. Unadjusted outcomes. In cases of missing data, sample sizes are indicated at bottom of graph. Clinical response = reduction in Harvey-Bradshaw Index (HBI) by >3 pts. Steroid-free clinical remission = HBI <5 with no use of corticosteroids at follow-up. Endoscopic response = simple endoscopic score for CD (SES-CD) reduction of 50%. Radiologic response = improved CD findings without new findings of active CD per radiologist’s assessment of cross-sectional imaging. Reference endoscopy/imaging studies were required to be within 1 year prior to treatment initiation. Provider and endoscopist global assessments were utilized when HBI and SES-CD were not documented or applicable. B and C. Competing risk regression analyses with weighted cumulative incidence curves for treatment discontinuation due to non-response for vedolizumab vs upadacitinib (B) and vedolizumab vs risankizumab (C). Curves are adjusted for treatment discontinuation due to adverse events as a competing event. D and E. IPTW logistic and Cox regression analysis results for vedolizumab vs upadacitinib (D) and vedolizumab vs risankizumab (E).
Abbreviations: OR=odds ratio, HR=hazard ratio
Disclosures:
Rahul Dalal: Janssen – Grant/Research Support. Takeda – Consultant.
Grace Hardwick indicated no relevant financial relationships.
Lindsay Clarke indicated no relevant financial relationships.
Heidy Cabral indicated no relevant financial relationships.
Alexander Carlin indicated no relevant financial relationships.
Jessica Allegretti: Abbvie – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speaker, Speakers Bureau. Adiso – Consultant. Bristol Myer Squibb – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speaker. Celltrion – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Ferring – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Finch – Consultant. Genentech – Advisory Committee/Board Member, Consultant. GlaxoSmithKline – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Iterative Scopes – Consultant. Janssen – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Speakers Bureau. Johnson & Johnson – Consultant, Grant/Research Support, Speaker. Merck – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Grant/Research Support. Pfizer – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant, Grant/Research Support. Roivant – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Roivant Adiso – Consultant. Seres Therapeutics – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Shattuck Labs – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. TRXBio – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant. Vedanta – Advisor or Review Panel Member, Advisory Committee/Board Member, Consultant.
Rahul S. Dalal, MD, MPH1, Grace Hardwick, BS1, Lindsay Clarke, MD1, Heidy Cabral, MS, BS1, Alexander Carlin, BS1, Jessica R.. Allegretti, MD, MPH2, 71, Real-World Comparative Effectiveness of Vedolizumab versus Upadacitinib and Risankizumab for Crohn’s Disease Through 52 Weeks, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.

