Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
66 - Duvakitug, an anti-TL1a mAb, Demonstrates Efficacy and Favorable Safety as an Induction Treatment in Adults With Moderately to Severely Active Crohn’s Disease: Results From the RELIEVE UCCD Phase 2b Basket Trial
Wednesday, October 29, 2025
8:40 AM - 8:50 AM PDT
Location: North Ballroom 120BC

Vipul Jairath, MBChB, DPhil, MRCP
Department of Medicine and Department of Epidemiology and Biostatistics, Western University
London, ON, Canada
Presenting Author(s)
Vipul Jairath, MBChB, DPhil, MRCP1, Jarosław Kierkuś, MD2, Aaron Duvall, MD3, Silvio Danese, MD, PhD4, Bruce E.. Sands, MD, MS, FACG5, Bogdan Ratiu-Duma, 6, Rajendra Singh, PhD7, Hadas Barkay, PhD8, Gordon Raphael, MD9, Walter Reinisch, MD, PhD10
1Western University, London, ON, Canada; 2The Children’s Memorial Health Institute, Warsaw, Mazowieckie, Poland; 3Tyler Research Institute, Tyler, TX; 4IRCCS Ospedale San Raffaele and University Vita-Salute San Raffaele, Milan, Lombardia, Italy; 5Mount Sinai, New York, NY; 6Teva Pharmaceuticals S.R.L., Pharmacovigilance, Bucharest, Bucuresti, Romania; 7Teva Branded Pharmaceutical Products, Research and Development, West Chester, PA; 8Teva Pharmaceutical Industries Ltd., Research and Development, Netanya, HaDarom, Israel; 9Teva Pharmaceutical Industries Ltd, West Chester, PA; 10Medical University of Vienna, Spitalgasse, Wien, Austria
Introduction: Duvakitug is a human IgG1 monoclonal antibody selected for its preferential inhibition of the TL1A-DR3 signaling over DcR3 binding. Duvakitug has demonstrated reduced inflammation and fibrosis in colitis animal models.1 Few data are available for the potential of anti-TL1A therapies in Crohn’s disease (CD). A phase 2b induction basket trial (NCT05499130) assessed the efficacy, safety and tolerability of duvakitug in adults with moderately to severely active ulcerative colitis and CD.
Methods: RELIEVE UCCD was a randomized, placebo (PBO)-controlled, double-blind induction study. The CD cohort comprised of adults with moderately to severely active disease with documented inadequate response, loss of response or intolerance to conventional and/or advanced therapies (ATs). Patients were randomized to receive subcutaneously a 2250 mg loading dose of duvakitug or PBO, followed by either duvakitug 450 mg, 900 mg or PBO (1:1:1; stratified by prior AT) every 2 weeks. Primary endpoint was endoscopic response (≥50% reduction from baseline in Simple Endoscopic Score for CD [SES-CD]) at week 14. Safety and tolerability were assessed by adverse event (AE) reporting and laboratory monitoring.
Results: In total, 138 patients with CD were randomized, treated and included in the analysis (n=46 per arm). Demographics and baseline characteristics were similar across arms (Table 1). Both duvakitug doses achieved the primary endpoint (26% [450 mg], 48% [900 mg] vs 13% [PBO]; PBO-adjusted rates: 13% [450 mg], 35% [900 mg]) with statistically significant endoscopic responses based on the prespecified Bayesian analysis, with a >0.90 posterior probability that each duvakitug dose is superior to PBO. Duvakitug treatment effect was observed in both AT-experienced and -naïve patients (Table 2). AE incidence was similar for duvakitug 900 mg (43%) and PBO (48%), and lower than duvakitug 450 mg (67%). Incidences of AEs leading to discontinuation were 2% each for duvakitug 900 mg and PBO, and 9% for duvakitug 450 mg.
Discussion: These are the first data reported for a double-blind, randomized, PBO-controlled induction study of an anti-TL1A antibody in patients with CD. Duvakitug demonstrated statistically significant and clinically meaningful endoscopic response versus PBO with no emergent safety signals observed. These results support further development of duvakitug as a treatment option for patients with moderately to severely active CD.
Reference:
1. Clarke AW, et al. MAbs. 2018;10:664–77
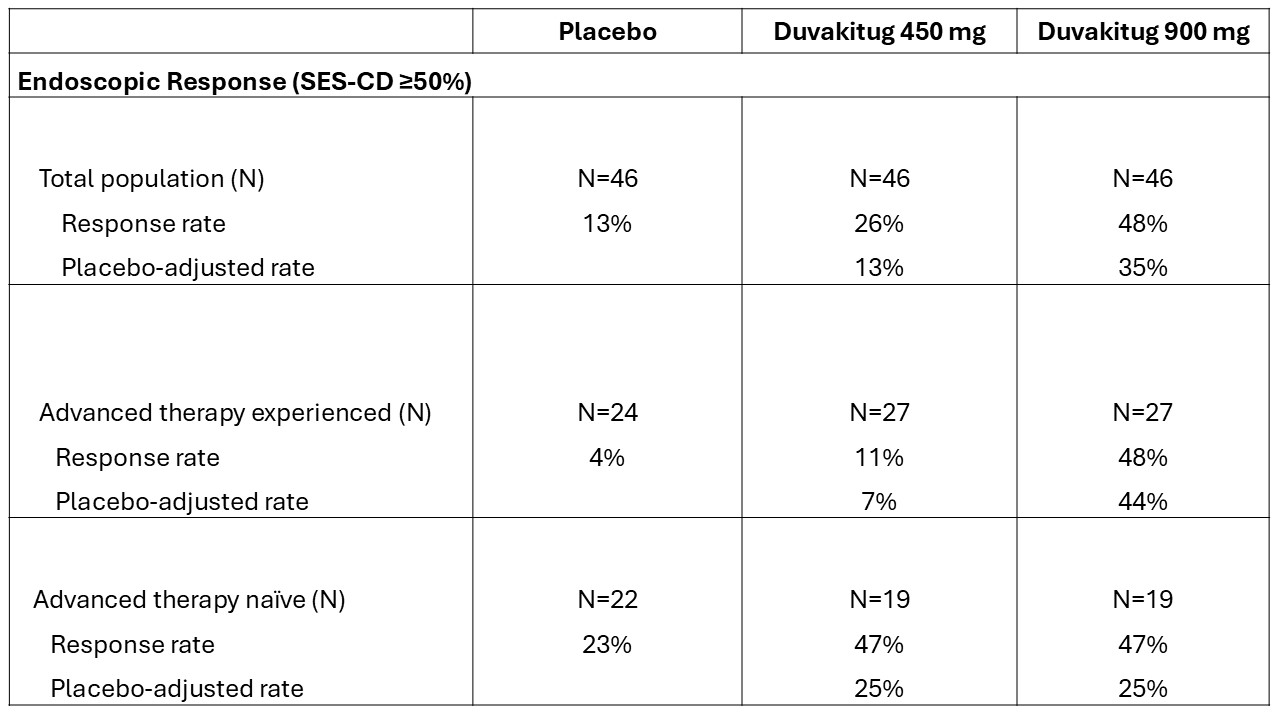
Figure: Table 1: Demographics and baseline characteristics.
Advanced therapies include approved therapies: biologics (TNF inhibitors, integrins inhibitors, IL-12/23 inhibitors or anti-IL-23), JAK inhibitors and S1P receptor modulators. Drugs currently in development for IBD are included in the investigational drugs category. Percentages may not add up to 100 due to rounding.
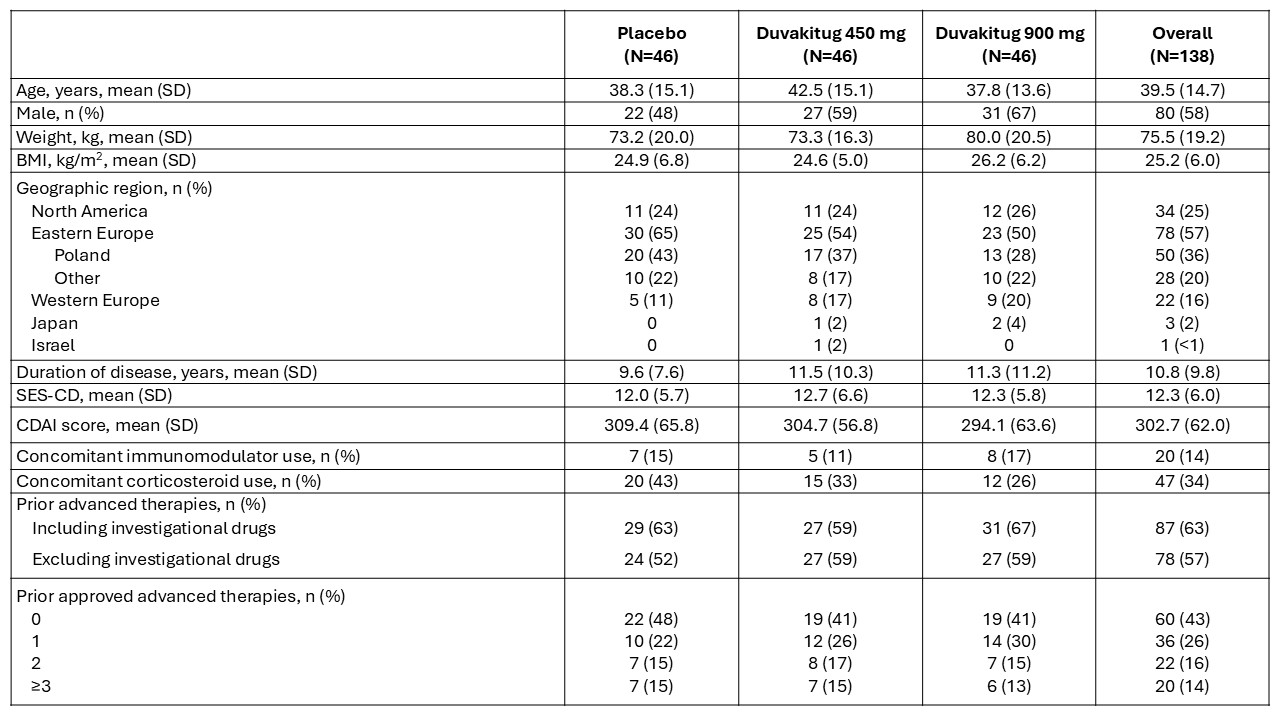
Figure: Table 2: Week 14 endoscopic response rates (SES-CD).
Advanced therapy experience includes prior exposure to approved biologics, S1P receptor modulators and/or JAK inhibitors. Percentages may not add up due to rounding.
Disclosures:
Vipul Jairath: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Alimentiv – Consultant, Grant/Research Support. Arena Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Asahi Kasei Pharma – Consultant, Grant/Research Support, Speakers Bureau. Asieris Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. AstraZeneca – Consultant, Grant/Research Support, Speakers Bureau. Avoro Capital – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, Speakers Bureau. Celltrion – Consultant, Grant/Research Support, Speakers Bureau. Eli Lilly and Company – Consultant, Grant/Research Support, Speakers Bureau. Endpoint Health – Consultant. Enthera – Consultant. Ferring Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Flagship Pioneering – Consultant, Grant/Research Support, Speakers Bureau. Fresenius Kabi – Consultant, Grant/Research Support, Speakers Bureau. Galapagos NV – Consultant, Grant/Research Support, Speakers Bureau. Genentech – Consultant, Grant/Research Support, Speakers Bureau. Gilde Healthcare – Consultant. Gilead Sciences – Consultant, Grant/Research Support, Speakers Bureau. GlaxoSmithKline – Consultant, Grant/Research Support, Speakers Bureau. Innomar – Consultant. JAMP – Consultant. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Merck – Consultant, Grant/Research Support, Speakers Bureau. Metacrine – Consultant, Grant/Research Support, Speakers Bureau. Mylan – Consultant, Grant/Research Support, Speakers Bureau. Pandion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Pendopharm – Consultant, Grant/Research Support, Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. Prometheus Therapeutics and Diagnostics – Consultant, Grant/Research Support, Speakers Bureau. Protagonist Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Reistone Biopharma – Consultant, Grant/Research Support, Speakers Bureau. Roche – Consultant, Grant/Research Support, Speakers Bureau. Roivant – Consultant. Sandoz – Consultant, Grant/Research Support, Speakers Bureau. Second Genome – Consultant, Grant/Research Support, Speakers Bureau. Shire – Speakers Bureau. Sorriso Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Syndegen – Consultant. Takeda – Consultant, Intellectual Property/Patents, Speakers Bureau. TD Securities – Consultant. Teva – Consultant, Grant/Research Support, Speakers Bureau. Topivert – Consultant, Grant/Research Support, Speakers Bureau. Ventyx Biosciences – Consultant, Grant/Research Support, Speakers Bureau. Vividion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau.
Jarosław Kierkuś indicated no relevant financial relationships.
Aaron Duvall indicated no relevant financial relationships.
Silvio Danese: AbbVie – Consultant, Lecture fees. Alimentiv – Consultant. Allergan – Consultant. Amgen – Consultant, Lecture fees. AstraZeneca – Consultant. Athos Therapeutics – Consultant. Biogen – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Celltrion – Consultant. Eli Lilly – Consultant. Enthera – Consultant. F. Hoffmann-La Roche Ltd – Consultant. Ferring Pharmaceuticals Inc. – Consultant, Lecture fees. Gilead – Consultant, Lecture fees. Hospira – Consultant. Inotrem – Consultant. Johnson & Johnson – Consultant, Lecture fees. MSD – Consultant. Mundipharma – Consultant. Mylan – Consultant, Lecture fees. Pfizer – Consultant, Lecture fees. Sandoz – Consultant. Sublimity Therapeutics – Consultant. Takeda – Consultant, Lecture fees. TiGenix – Consultant. UCB Inc. – Consultant. Vifor (International) Ltd. – Consultant.
Bruce Sands: AbbVie – Consultant. Abivax – Consultant. Agomab Therapeutics – Consultant. Alimentiv – Consultant. Amgen – Consultant. AnaptysBio – Consultant. Boehringer Ingelheim – Consultant. Celltrion – Consultant, Editorial Support. Eli Lilly and Company – Consultant, Editorial Support. Ferring Pharmaceuticals – Consultant. Galapagos – Consultant. Genentech (Roche) – Consultant, Editorial Support. Gilead Sciences – Consultant. Janssen R&D – Consultant, Grant/Research Support, Editorial Support. Johnson & Johnson Innovative Medicine – Consultant, Editorial Support. Merck & Co. – Consultant, Editorial Support. Pfizer – Consultant, Editorial Support. Sanofi – Consultant. Takeda – Consultant, Editorial Support. Target RWE – Consultant. Teva – Consultant, Editorial Support. Ventyx Biosciences – Consultant, Editorial Support, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Bogdan Ratiu-Duma: Teva – Employee.
Rajendra Singh: Teva – Employee.
Hadas Barkay: Teva – Employee.
Gordon Raphael: Teva – Employee.
Walter Reinisch: AbbVie – Advisory Committee/Board Member, Consultant, Grant/Research Support. Actelion – Advisory Committee/Board Member, Consultant. Alpha Wasserman – Advisory Committee/Board Member, Consultant. AstraZeneca – Advisory Committee/Board Member, Consultant. Cellerix – Advisory Committee/Board Member, Consultant. Cosmo Pharmaceuticals – Advisory Committee/Board Member, Consultant. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Grunenthal – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant. Merck – Advisory Committee/Board Member, Consultant. Millennium – Advisory Committee/Board Member, Consultant. Novo Nordisk – Advisory Committee/Board Member, Consultant. Nycomed – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Pharmacosmos – Advisory Committee/Board Member, Consultant. Salix Pharmaceuticals – Advisory Committee/Board Member, Consultant. Schering-Plough – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB Pharma – Advisory Committee/Board Member, Consultant. Vifor Pharma – Advisory Committee/Board Member, Consultant.
Vipul Jairath, MBChB, DPhil, MRCP1, Jarosław Kierkuś, MD2, Aaron Duvall, MD3, Silvio Danese, MD, PhD4, Bruce E.. Sands, MD, MS, FACG5, Bogdan Ratiu-Duma, 6, Rajendra Singh, PhD7, Hadas Barkay, PhD8, Gordon Raphael, MD9, Walter Reinisch, MD, PhD10, 66, Duvakitug, an anti-TL1a mAb, Demonstrates Efficacy and Favorable Safety as an Induction Treatment in Adults With Moderately to Severely Active Crohn’s Disease: Results From the RELIEVE UCCD Phase 2b Basket Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.
1Western University, London, ON, Canada; 2The Children’s Memorial Health Institute, Warsaw, Mazowieckie, Poland; 3Tyler Research Institute, Tyler, TX; 4IRCCS Ospedale San Raffaele and University Vita-Salute San Raffaele, Milan, Lombardia, Italy; 5Mount Sinai, New York, NY; 6Teva Pharmaceuticals S.R.L., Pharmacovigilance, Bucharest, Bucuresti, Romania; 7Teva Branded Pharmaceutical Products, Research and Development, West Chester, PA; 8Teva Pharmaceutical Industries Ltd., Research and Development, Netanya, HaDarom, Israel; 9Teva Pharmaceutical Industries Ltd, West Chester, PA; 10Medical University of Vienna, Spitalgasse, Wien, Austria
Introduction: Duvakitug is a human IgG1 monoclonal antibody selected for its preferential inhibition of the TL1A-DR3 signaling over DcR3 binding. Duvakitug has demonstrated reduced inflammation and fibrosis in colitis animal models.1 Few data are available for the potential of anti-TL1A therapies in Crohn’s disease (CD). A phase 2b induction basket trial (NCT05499130) assessed the efficacy, safety and tolerability of duvakitug in adults with moderately to severely active ulcerative colitis and CD.
Methods: RELIEVE UCCD was a randomized, placebo (PBO)-controlled, double-blind induction study. The CD cohort comprised of adults with moderately to severely active disease with documented inadequate response, loss of response or intolerance to conventional and/or advanced therapies (ATs). Patients were randomized to receive subcutaneously a 2250 mg loading dose of duvakitug or PBO, followed by either duvakitug 450 mg, 900 mg or PBO (1:1:1; stratified by prior AT) every 2 weeks. Primary endpoint was endoscopic response (≥50% reduction from baseline in Simple Endoscopic Score for CD [SES-CD]) at week 14. Safety and tolerability were assessed by adverse event (AE) reporting and laboratory monitoring.
Results: In total, 138 patients with CD were randomized, treated and included in the analysis (n=46 per arm). Demographics and baseline characteristics were similar across arms (Table 1). Both duvakitug doses achieved the primary endpoint (26% [450 mg], 48% [900 mg] vs 13% [PBO]; PBO-adjusted rates: 13% [450 mg], 35% [900 mg]) with statistically significant endoscopic responses based on the prespecified Bayesian analysis, with a >0.90 posterior probability that each duvakitug dose is superior to PBO. Duvakitug treatment effect was observed in both AT-experienced and -naïve patients (Table 2). AE incidence was similar for duvakitug 900 mg (43%) and PBO (48%), and lower than duvakitug 450 mg (67%). Incidences of AEs leading to discontinuation were 2% each for duvakitug 900 mg and PBO, and 9% for duvakitug 450 mg.
Discussion: These are the first data reported for a double-blind, randomized, PBO-controlled induction study of an anti-TL1A antibody in patients with CD. Duvakitug demonstrated statistically significant and clinically meaningful endoscopic response versus PBO with no emergent safety signals observed. These results support further development of duvakitug as a treatment option for patients with moderately to severely active CD.
Reference:
1. Clarke AW, et al. MAbs. 2018;10:664–77

Figure: Table 1: Demographics and baseline characteristics.
Advanced therapies include approved therapies: biologics (TNF inhibitors, integrins inhibitors, IL-12/23 inhibitors or anti-IL-23), JAK inhibitors and S1P receptor modulators. Drugs currently in development for IBD are included in the investigational drugs category. Percentages may not add up to 100 due to rounding.

Figure: Table 2: Week 14 endoscopic response rates (SES-CD).
Advanced therapy experience includes prior exposure to approved biologics, S1P receptor modulators and/or JAK inhibitors. Percentages may not add up due to rounding.
Disclosures:
Vipul Jairath: AbbVie – Consultant, Grant/Research Support, Speakers Bureau. Alimentiv – Consultant, Grant/Research Support. Arena Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Asahi Kasei Pharma – Consultant, Grant/Research Support, Speakers Bureau. Asieris Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. AstraZeneca – Consultant, Grant/Research Support, Speakers Bureau. Avoro Capital – Consultant. Bristol Myers Squibb – Consultant, Grant/Research Support, Speakers Bureau. Celltrion – Consultant, Grant/Research Support, Speakers Bureau. Eli Lilly and Company – Consultant, Grant/Research Support, Speakers Bureau. Endpoint Health – Consultant. Enthera – Consultant. Ferring Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Flagship Pioneering – Consultant, Grant/Research Support, Speakers Bureau. Fresenius Kabi – Consultant, Grant/Research Support, Speakers Bureau. Galapagos NV – Consultant, Grant/Research Support, Speakers Bureau. Genentech – Consultant, Grant/Research Support, Speakers Bureau. Gilde Healthcare – Consultant. Gilead Sciences – Consultant, Grant/Research Support, Speakers Bureau. GlaxoSmithKline – Consultant, Grant/Research Support, Speakers Bureau. Innomar – Consultant. JAMP – Consultant. Johnson & Johnson – Advisor or Review Panel Member, Speakers Bureau. Merck – Consultant, Grant/Research Support, Speakers Bureau. Metacrine – Consultant, Grant/Research Support, Speakers Bureau. Mylan – Consultant, Grant/Research Support, Speakers Bureau. Pandion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Pendopharm – Consultant, Grant/Research Support, Speakers Bureau. Pfizer – Consultant, Grant/Research Support, Speakers Bureau. Prometheus Therapeutics and Diagnostics – Consultant, Grant/Research Support, Speakers Bureau. Protagonist Therapeutics – Consultant, Grant/Research Support, Speakers Bureau. Reistone Biopharma – Consultant, Grant/Research Support, Speakers Bureau. Roche – Consultant, Grant/Research Support, Speakers Bureau. Roivant – Consultant. Sandoz – Consultant, Grant/Research Support, Speakers Bureau. Second Genome – Consultant, Grant/Research Support, Speakers Bureau. Shire – Speakers Bureau. Sorriso Pharmaceuticals – Consultant, Grant/Research Support, Speakers Bureau. Syndegen – Consultant. Takeda – Consultant, Intellectual Property/Patents, Speakers Bureau. TD Securities – Consultant. Teva – Consultant, Grant/Research Support, Speakers Bureau. Topivert – Consultant, Grant/Research Support, Speakers Bureau. Ventyx Biosciences – Consultant, Grant/Research Support, Speakers Bureau. Vividion Therapeutics – Consultant, Grant/Research Support, Speakers Bureau.
Jarosław Kierkuś indicated no relevant financial relationships.
Aaron Duvall indicated no relevant financial relationships.
Silvio Danese: AbbVie – Consultant, Lecture fees. Alimentiv – Consultant. Allergan – Consultant. Amgen – Consultant, Lecture fees. AstraZeneca – Consultant. Athos Therapeutics – Consultant. Biogen – Consultant. Boehringer Ingelheim – Consultant. Celgene – Consultant. Celltrion – Consultant. Eli Lilly – Consultant. Enthera – Consultant. F. Hoffmann-La Roche Ltd – Consultant. Ferring Pharmaceuticals Inc. – Consultant, Lecture fees. Gilead – Consultant, Lecture fees. Hospira – Consultant. Inotrem – Consultant. Johnson & Johnson – Consultant, Lecture fees. MSD – Consultant. Mundipharma – Consultant. Mylan – Consultant, Lecture fees. Pfizer – Consultant, Lecture fees. Sandoz – Consultant. Sublimity Therapeutics – Consultant. Takeda – Consultant, Lecture fees. TiGenix – Consultant. UCB Inc. – Consultant. Vifor (International) Ltd. – Consultant.
Bruce Sands: AbbVie – Consultant. Abivax – Consultant. Agomab Therapeutics – Consultant. Alimentiv – Consultant. Amgen – Consultant. AnaptysBio – Consultant. Boehringer Ingelheim – Consultant. Celltrion – Consultant, Editorial Support. Eli Lilly and Company – Consultant, Editorial Support. Ferring Pharmaceuticals – Consultant. Galapagos – Consultant. Genentech (Roche) – Consultant, Editorial Support. Gilead Sciences – Consultant. Janssen R&D – Consultant, Grant/Research Support, Editorial Support. Johnson & Johnson Innovative Medicine – Consultant, Editorial Support. Merck & Co. – Consultant, Editorial Support. Pfizer – Consultant, Editorial Support. Sanofi – Consultant. Takeda – Consultant, Editorial Support. Target RWE – Consultant. Teva – Consultant, Editorial Support. Ventyx Biosciences – Consultant, Editorial Support, Stock Options, Stock-publicly held company(excluding mutual/index funds).
Bogdan Ratiu-Duma: Teva – Employee.
Rajendra Singh: Teva – Employee.
Hadas Barkay: Teva – Employee.
Gordon Raphael: Teva – Employee.
Walter Reinisch: AbbVie – Advisory Committee/Board Member, Consultant, Grant/Research Support. Actelion – Advisory Committee/Board Member, Consultant. Alpha Wasserman – Advisory Committee/Board Member, Consultant. AstraZeneca – Advisory Committee/Board Member, Consultant. Cellerix – Advisory Committee/Board Member, Consultant. Cosmo Pharmaceuticals – Advisory Committee/Board Member, Consultant. Ferring Pharmaceuticals – Advisory Committee/Board Member, Consultant. Genentech – Advisory Committee/Board Member, Consultant. Grunenthal – Advisory Committee/Board Member, Consultant. Johnson & Johnson – Advisory Committee/Board Member, Consultant. Merck – Advisory Committee/Board Member, Consultant. Millennium – Advisory Committee/Board Member, Consultant. Novo Nordisk – Advisory Committee/Board Member, Consultant. Nycomed – Advisory Committee/Board Member, Consultant. Pfizer – Advisory Committee/Board Member, Consultant. Pharmacosmos – Advisory Committee/Board Member, Consultant. Salix Pharmaceuticals – Advisory Committee/Board Member, Consultant. Schering-Plough – Advisory Committee/Board Member, Consultant. Takeda – Advisory Committee/Board Member, Consultant. UCB Pharma – Advisory Committee/Board Member, Consultant. Vifor Pharma – Advisory Committee/Board Member, Consultant.
Vipul Jairath, MBChB, DPhil, MRCP1, Jarosław Kierkuś, MD2, Aaron Duvall, MD3, Silvio Danese, MD, PhD4, Bruce E.. Sands, MD, MS, FACG5, Bogdan Ratiu-Duma, 6, Rajendra Singh, PhD7, Hadas Barkay, PhD8, Gordon Raphael, MD9, Walter Reinisch, MD, PhD10, 66, Duvakitug, an anti-TL1a mAb, Demonstrates Efficacy and Favorable Safety as an Induction Treatment in Adults With Moderately to Severely Active Crohn’s Disease: Results From the RELIEVE UCCD Phase 2b Basket Trial, ACG 2025 Annual Scientific Meeting Abstracts. Phoenix, AZ: American College of Gastroenterology.


