Oral Paper Presentation
Annual Scientific Meeting
Session: Plenary Session 4B: IBD
74 - AVB-114 Demonstrates Clinical Benefit in Persistent Crohn’s Perianal Fistulas: Results From the Multicenter Randomized STOMP2 Trial (Late-Breaking Abstract)
Wednesday, October 29, 2025
10:00 AM - 10:10 AM PDT
Location: North Ballroom 120BC

David A. Schwartz, MD, FACG (he/him/his)
Professor of Medicine
Vanderbilt University Medical Center
Nashville, TN
Late Breaking Abstract Presenter(s)
David A. Schwartz, MD, FACG1, Eric Dozois, MD2, Eric Ehman, MD3, William Faubion, MD4, David P. Hudesman, MD, FACG5, Todd Johnson, PhD6, Baldeep Pabla, MD, MSCI1, Konstantinos A. Papadakis, MD, PhD3, Phillip Fleshner, MD71Vanderbilt University Medical Center, Brentwood, TN; 2Mayo Clinic College of Medicine and Science, Rochester, MN; 3Mayo Clinic, Rochester, MN; 4Mayo Clinic, Phoenix, AZ; 5NYU Langone Health, New York, NY; 6Avobis Bio, LLC, Newark, DE; 7Cedars-Sinai Medical Center, Los Angeles, CA
Introduction: Nearly a quarter of patients with Crohn’s disease (CD) will develop a perianal fistula. Despite the use of advanced therapies, the majority of these pts will not achieve complete closure of their fistula tracts. The primary objective of the STOMP2 clinical trial was to determine whether the implantable autologous cell therapy AVB-114 is effective in inducing remission of persistent Crohn’s perianal fistulas versus standard of care.
Methods: Eligible patients had a single perianal fistula tract and had failed prior treatment or had documented medication intolerance to biologic or conventional CD therapy. Prior to treatment, all enrolled subjects received surgical fistula optimization (including debridement and seton placement) and had an adipose tissue biopsy (in order to manufacture AVB-114). Subjects were subsequently randomized 1:1 to either standard of care (repeat fistula optimization including seton replacement) or AVB-114 implantation. The primary endpoint was combined fistula remission at 9 months. Remission was defined as closure of the external opening, no drainage of fluid despite gentle finger compression, and no collections >2cm in at least two of the three dimensions on MRI. Secondary endpoints included clinical remission, pt-reported outcome measures, and radiological assessments.
Results: Subjects at 14 U.S. sites were equally randomized to SoC (N=24) or AVB-114 (N=24) (Table 1). At 9 mo, combined remission was 8.3% for SoC and 45.8% for AVB-114 (38% difference, 95% CI 11-60%; p=0.0078), with key secondary endpoints showing improvement with AVB-114 (Figure 1 and Table 2). In Part 1 through 9 mo, there were 40 and 56 treatment emergent adverse events (TEAE) for SoC and AVB-114, respectively. The increase in AVB-114 events vs SoC was predominantly due to non-serious postoperative perianal pain. There was one serious TEAE in AVB-114 (pseudopolyposis) assessed as “not related” to the investigational product. Two grade 2 (moderate) AEs of cellulitis and abdominal wall abscess were attributed to adipose tissue collection (subject disposition of screen failure and SoC treatment, respectively).
Discussion: A significantly higher proportion of patients treated with AVB-114 achieved combined remission versus SoC. Improvements in secondary endpoints were observed through 9 months, and the treatment was well tolerated. These data supports the safety and efficacy of AVB-114 in refractory Crohn’s perianal fistulas.
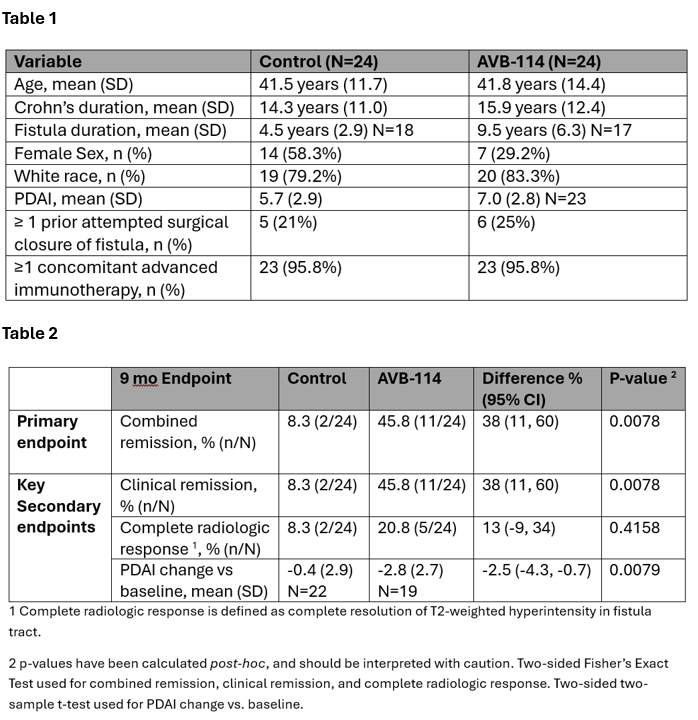
Figure: Table 1: Demographics and medical/surgical history Table 2: Primary and key secondary analysis through 9 months
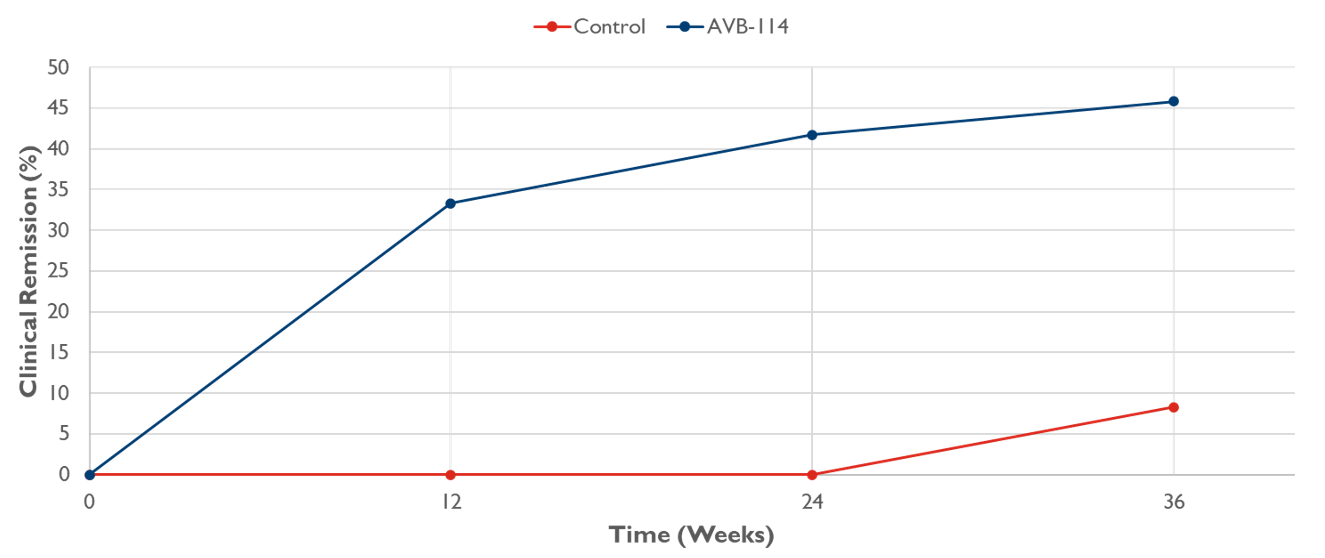
Figure: Figure 1: Clinical remission through 9 months
Disclosures:
David Schwartz: Abbvie – Consultant, Speakers Bureau. Avobis – Advisory Committee/Board Member. janssen – Advisory Committee/Board Member, Consultant, Speakers Bureau. olympus – Advisory Committee/Board Member, Consultant. Takeda – Consultant.
Eric Dozois: Avobis Bio – Grant/Research Support, Intellectual Property/Patents.
Eric Ehman indicated no relevant financial relationships.
William Faubion indicated no relevant financial relationships.
David Hudesman: Abbvie – Consultant. Abivax – Consultant. Avalo – Consultant. Biocon – Consultant. BMS – Consultant. CorEvitas – Consultant. Eli Lilly – Consultant. Fresenius Kabi – Consultant. Johnson and Johnson – Consultant. Pfizer – Consultant, Grant/Research Support. Prometheus – Consultant. Sanofi – Consultant. Takeda – Consultant.
Todd Johnson: Avobis Bio, LLC – Employee.
Baldeep Pabla: Abbvie – Advisor or Review Panel Member. Astellas – Advisory Committee/Board Member. Eli Lilly – Speakers Bureau. J&J – Advisory Committee/Board Member. Prometheus – Speakers Bureau.
Konstantinos Papadakis indicated no relevant financial relationships.
Phillip Fleshner: avobis bio – Consultant. olympus – Consultant. takeda – Consultant.
Introduction: Nearly a quarter of patients with Crohn’s disease (CD) will develop a perianal fistula. Despite the use of advanced therapies, the majority of these pts will not achieve complete closure of their fistula tracts. The primary objective of the STOMP2 clinical trial was to determine whether the implantable autologous cell therapy AVB-114 is effective in inducing remission of persistent Crohn’s perianal fistulas versus standard of care.
Methods: Eligible patients had a single perianal fistula tract and had failed prior treatment or had documented medication intolerance to biologic or conventional CD therapy. Prior to treatment, all enrolled subjects received surgical fistula optimization (including debridement and seton placement) and had an adipose tissue biopsy (in order to manufacture AVB-114). Subjects were subsequently randomized 1:1 to either standard of care (repeat fistula optimization including seton replacement) or AVB-114 implantation. The primary endpoint was combined fistula remission at 9 months. Remission was defined as closure of the external opening, no drainage of fluid despite gentle finger compression, and no collections >2cm in at least two of the three dimensions on MRI. Secondary endpoints included clinical remission, pt-reported outcome measures, and radiological assessments.
Results: Subjects at 14 U.S. sites were equally randomized to SoC (N=24) or AVB-114 (N=24) (Table 1). At 9 mo, combined remission was 8.3% for SoC and 45.8% for AVB-114 (38% difference, 95% CI 11-60%; p=0.0078), with key secondary endpoints showing improvement with AVB-114 (Figure 1 and Table 2). In Part 1 through 9 mo, there were 40 and 56 treatment emergent adverse events (TEAE) for SoC and AVB-114, respectively. The increase in AVB-114 events vs SoC was predominantly due to non-serious postoperative perianal pain. There was one serious TEAE in AVB-114 (pseudopolyposis) assessed as “not related” to the investigational product. Two grade 2 (moderate) AEs of cellulitis and abdominal wall abscess were attributed to adipose tissue collection (subject disposition of screen failure and SoC treatment, respectively).
Discussion: A significantly higher proportion of patients treated with AVB-114 achieved combined remission versus SoC. Improvements in secondary endpoints were observed through 9 months, and the treatment was well tolerated. These data supports the safety and efficacy of AVB-114 in refractory Crohn’s perianal fistulas.

Figure: Table 1: Demographics and medical/surgical history Table 2: Primary and key secondary analysis through 9 months

Figure: Figure 1: Clinical remission through 9 months
Disclosures:
David Schwartz: Abbvie – Consultant, Speakers Bureau. Avobis – Advisory Committee/Board Member. janssen – Advisory Committee/Board Member, Consultant, Speakers Bureau. olympus – Advisory Committee/Board Member, Consultant. Takeda – Consultant.
Eric Dozois: Avobis Bio – Grant/Research Support, Intellectual Property/Patents.
Eric Ehman indicated no relevant financial relationships.
William Faubion indicated no relevant financial relationships.
David Hudesman: Abbvie – Consultant. Abivax – Consultant. Avalo – Consultant. Biocon – Consultant. BMS – Consultant. CorEvitas – Consultant. Eli Lilly – Consultant. Fresenius Kabi – Consultant. Johnson and Johnson – Consultant. Pfizer – Consultant, Grant/Research Support. Prometheus – Consultant. Sanofi – Consultant. Takeda – Consultant.
Todd Johnson: Avobis Bio, LLC – Employee.
Baldeep Pabla: Abbvie – Advisor or Review Panel Member. Astellas – Advisory Committee/Board Member. Eli Lilly – Speakers Bureau. J&J – Advisory Committee/Board Member. Prometheus – Speakers Bureau.
Konstantinos Papadakis indicated no relevant financial relationships.
Phillip Fleshner: avobis bio – Consultant. olympus – Consultant. takeda – Consultant.

